Journal Description
Separations
Separations
- formerly Chromatography - is an international, scientific, peer-reviewed, open access journal on separation and purification science and technology in all areas of chemical, biological, physical science, and separation performance published monthly online by MDPI. The Central European Group of Separation Sciences (CEGSS) is affiliated with Separations and their members receive a discount on the article processing charges.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, SCIE (Web of Science), CAPlus / SciFinder, and other databases.
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 12.9 days after submission; acceptance to publication is undertaken in 2.9 days (median values for papers published in this journal in the first half of 2023).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
Impact Factor:
2.6 (2022);
5-Year Impact Factor:
2.7 (2022)
Latest Articles
In-Line Measurement of Extraction Process by Slug Flow and Determination of Mass Transfer Parameters
Separations 2023, 10(8), 443; https://doi.org/10.3390/separations10080443 - 08 Aug 2023
Abstract
The primary objective of this study was to pragmatically implement an extraction process using slug flow, wherein two immiscible phases, aqueous and oil, are alternatively channeled through a conduit to encourage mass exchange across the interface. To facilitate this, we introduced and empirically
[...] Read more.
The primary objective of this study was to pragmatically implement an extraction process using slug flow, wherein two immiscible phases, aqueous and oil, are alternatively channeled through a conduit to encourage mass exchange across the interface. To facilitate this, we introduced and empirically validated an in-line technique to precisely quantify the length of the slug and the potential extraction concentration of the two aforementioned phases. The length of the slug and its concentration were ascertained through conductivity, utilizing platinum wire as the electrode. This method consistently produced results with a maximum error margin of ±5%. Using this apparatus, we determined key mass transfer parameters, including the overall mass transfer coefficient. Notably, as the linear velocity amplified, so did the extraction rate. These findings present a significant opportunity for enhancing the efficiency of the extraction process and enabling its optimization.
Full article
(This article belongs to the Topic Advances in Chemistry and Chemical Engineering)
►
Show Figures
Open AccessArticle
Effects of Catha edulis (Khat) on the Pharmacokinetics of Metformin in Diabetic Rats Using UPLC/MS/MS Analysis and Its Impact on Hepatic CYP450 Enzymes
by
, , , , , , , , and
Separations 2023, 10(8), 442; https://doi.org/10.3390/separations10080442 - 08 Aug 2023
Abstract
Catha edulis Forsk., commonly known as Khat, is a stimulant plant that is chewed in the Horn of Africa and the southern regions of the Arabian Peninsula. It is often used alongside conventional drugs, but there is a lack of research on
[...] Read more.
Catha edulis Forsk., commonly known as Khat, is a stimulant plant that is chewed in the Horn of Africa and the southern regions of the Arabian Peninsula. It is often used alongside conventional drugs, but there is a lack of research on its interactions with other drugs. In this study, our aim was to investigate the possibility of an herb–drug interaction between KT extract and metformin (MT) by exploring the effect of KT on the in vivo pharmacokinetics of MT in diabetic Wistar albino rats and monitoring blood glucose levels through time intervals, comparing when MT was given alone and co-administered with KT. Rats were given 500 mg/kg of KT extract and 300 mg/kg of MT daily for 7 days. The pharmacokinetic parameters of MT were analyzed using UPLC-MS/MS. The quantification was conducted using multiple reaction monitoring (MRM) in positive ion mode. The transitions used were (m/z) 130.006→59.939 for MT and 260.125→116.018 for propranolol as an internal standard (IS). An Acquity UPLC BEH HSS T3 C18 column was used with isocratic elution, and the mobile phase was formic acid (0.1%): acetonitrile (70:30 v/v) at a flow rate of 0.25 mL/min. In addition, the study delved into the in vitro impact of KT on the CYP3A4 enzyme, seeking to identify any potential disruptions in MT’s metabolism that could alter its pharmacokinetics. The effect of KT extract on CYP3A4 enzyme activity was investigated using fluorescence-based enzyme assays and a CYP3A4 Inhibitor Screening Kit. The results indicated that the combined treatment showed increased systemic exposure to MT, with Cmax and AUC levels increasing by 33.42% and 45.23%, respectively. Additionally, the combination treatment led to significantly lower fasting blood glucose (FBG) levels compared to the groups treated with MT alone. It was found that at a concentration of 500 µg/mL, the CYP3A4 enzyme activity was maximally suppressed by approximately 57%, while at 250 µg/mL, it was inhibited by almost 50%. In comparison, the standard Ketoconazole only inhibited CYP3A4 activity by approximately 58%. Therefore, the in vitro results suggest that KT extract has the potential to affect CYP3A4 activity at high doses. In conclusion, the combination treatment resulted in a significant increase in MT’s blood sugar-lowering effects. Therefore, it is important to be aware of this potential interaction and to monitor blood sugar levels closely when using KT alongside MT.
Full article
(This article belongs to the Section Chromatographic Separations)
►▼
Show Figures

Figure 1
Open AccessReview
Enhanced Adsorption of Chromate and Arsenate Ions from Contaminated Water with Emphasis on Polyethylenimine Modified Materials: A Review
Separations 2023, 10(8), 441; https://doi.org/10.3390/separations10080441 - 07 Aug 2023
Abstract
Heavy metals in water pose a great threat to human health, and water treatment remains a critical issue globally. Among various water treatment techniques, adsorption has been proven to be an excellent way to remove heavy metals from water due to its advantages,
[...] Read more.
Heavy metals in water pose a great threat to human health, and water treatment remains a critical issue globally. Among various water treatment techniques, adsorption has been proven to be an excellent way to remove heavy metals from water due to its advantages, like low cost, availability, profitability, ease of operation, and efficiency. Heavy metals such as chromium and arsenic are present in water in the form of anions, while many common adsorbents, such as biochar and activated carbons, cellulose, chitosan, clays, and silica, tend to acquire a negative surface charge. The surface charge of the adsorbent plays a major role in its interaction with heavy metal ions. Polyethylenimine is a polymer with a high cationic charge density. Modification of these adsorbents with polyethylenimine results in higher selectivity for these anions. The adsorption capacity is related to the efficiency of the introduction of polyethylenimine, which depends on the surface properties of the adsorbent. This review focuses on the effect of modification with polyethylenimine on the adsorption capacity of common adsorbents for the removal of heavy metal ions, such as chromate and arsenate, from water.
Full article
(This article belongs to the Special Issue Removal of Metals or Other Toxic Substances from Wastewaters by Natural Sorbents)
Open AccessArticle
Enhancement of Photocatalytic Rhodamine B Degradation over Magnesium–Manganese Baring Extracted Iron Oxalate from Converter Slag
by
, , , , , , and
Separations 2023, 10(8), 440; https://doi.org/10.3390/separations10080440 - 03 Aug 2023
Abstract
In this work, iron oxalate from converter slag (FeOX-Slag) was produced by extraction of iron from converter slag using oxalic acid, followed by photo-reduction. The FeOX-Slag sample was subjected to various characterization techniques, including X-ray diffraction (XRD), Raman spectroscopy, scanning electron microscopy with
[...] Read more.
In this work, iron oxalate from converter slag (FeOX-Slag) was produced by extraction of iron from converter slag using oxalic acid, followed by photo-reduction. The FeOX-Slag sample was subjected to various characterization techniques, including X-ray diffraction (XRD), Raman spectroscopy, scanning electron microscopy with energy-dispersive X-ray spectroscopy (SEM-EDX), ultraviolet–visible diffuse reflectance spectroscopy (UV-DRS), photoluminescence spectroscopy (PL), X-ray absorption near-edge structure spectroscopy (XANES), and X-ray photoelectron spectroscopy (XPS), in order to gain insights into its physicochemical properties. Also, to compare the photocatalytic activity of the FeOX-Slag, commercial iron oxide (Fe2O3) was used as a precursor to produce normal iron oxalate (FeOX-Fe2O3). The obtained FeOX-Slag was applied to the photocatalytic degradation of rhodamine B (RhB), a model organic contaminant in wastewater, compared with the FeOX-Fe2O3. Using the produced FeOX-Slag, we were able to degrade RhB more than 98% within 90 min at a reaction rate constant of about 3.6 times faster than FeOX-Fe2O3. Photoluminescence results confirmed the less recombination of the electron–hole pairs in FeOX-Slag, compared to FeOX-Fe2O3, which may be due to the defect structure of iron oxalate by guest metal impurities. The higher separation and transportation of photogenerated electron–hole pairs cause the enhancement of the degradation photocatalytic RhB degradation activity of the FeOX-Slag. In addition, The FeOX-Slag showed higher light absorption ability than FeOX-Fe2O3, resulting in the enhancement of the RhB degradation performance. Thus, the optical properties and the results from the activity tests led to the proposal that FeOX-Slag may be used in a photocatalytic degradation process for RhB under light irradiation.
Full article
(This article belongs to the Special Issue Pollutants Removal by Photocatalytic Degradation)
►▼
Show Figures
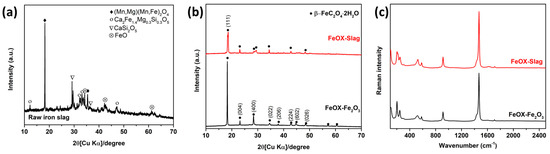
Figure 1
Open AccessEditorial
Chromatographic Separations
Separations 2023, 10(8), 439; https://doi.org/10.3390/separations10080439 - 02 Aug 2023
Abstract
The topic of chromatographic separations is a recent addition to the Separations journal [...]
Full article
(This article belongs to the Section Chromatographic Separations)
Open AccessArticle
Removal of Pb(II) from Aqueous Solution and Adsorption Kinetics of Corn Stalk Biochar
by
, , , , , , , , and
Separations 2023, 10(8), 438; https://doi.org/10.3390/separations10080438 - 02 Aug 2023
Abstract
In this work, the Pb adsorption and removal ability of biochar from simulated Pb(II)-contaminated wastewater, adsorption isotherms, kinetics, and thermodynamics were studied. Adsorption characteristics of biochar on Pb(II) were analyzed by Fourier transform infrared spectroscopy (FT-IR), X-ray diffraction (XRD) and scanning electron microscope
[...] Read more.
In this work, the Pb adsorption and removal ability of biochar from simulated Pb(II)-contaminated wastewater, adsorption isotherms, kinetics, and thermodynamics were studied. Adsorption characteristics of biochar on Pb(II) were analyzed by Fourier transform infrared spectroscopy (FT-IR), X-ray diffraction (XRD) and scanning electron microscope with energy dispersive spectrometer (SEM-EDS). The influence of the pH of the solution, the contact time, and the biochar dose on the removal of Pb(II) were investigated by single-factor design and response surface analysis. With the increase in biochar dose from 2 g/L to 4 g/L in wastewater, the Pb(II) amount adsorbed on biochar reduced from 21.3 mg/g to 17.5 mg/g. A weakly acidic environment was more conducive to the ligand exchange between Pb(II) ions and biochar. Pb(II) adsorption kinetics of biochar showed that the Pseudo-first-order model was more suitable than other employed models to describe the adsorption process. During the isothermal adsorption process, Langmuir and Freundlich’s isotherms fitted the adsorption data very well (R2 > 96%). The Pb (II) adsorption onto biochar was spontaneous in the specified temperature range (298–318 K) and the process was exothermic. Simultaneously, the optimal conditions were a pH of 5, a contact time of 255 min, and a biochar dose of 3 g/L, under which the maximum predicted Pb(II) removal efficiency was 91.52%.
Full article
(This article belongs to the Special Issue Carbon-Based Materials for Contaminant Removal)
►▼
Show Figures
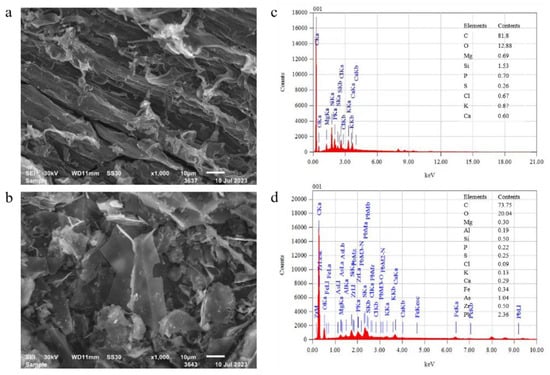
Figure 1
Open AccessArticle
Optimization of the Preparation of Hydrophilic Poly(DHPMA-co-MBA) Monolithic Capillary Columns: A New Support for Affinity Chromatography
by
, , , , and
Separations 2023, 10(8), 437; https://doi.org/10.3390/separations10080437 - 02 Aug 2023
Abstract
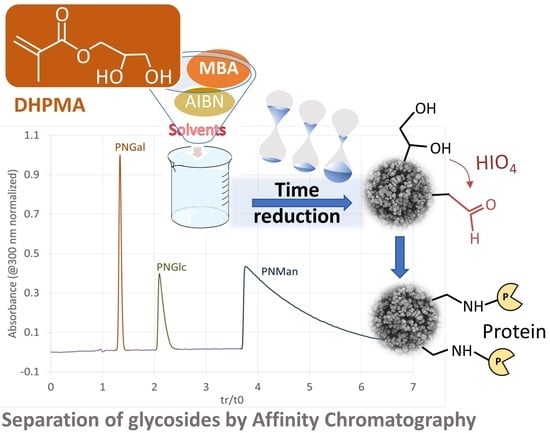
In miniaturized affinity chromatography, the development of hydrophilic organic monoliths with reduced non-specific interactions and high-protein grafting capacity remains a hot topic. In this work, we propose the one-step synthesis of a diol organic monolith to replace the gold-standard epoxy-based organic monoliths (which
[...] Read more.
In miniaturized affinity chromatography, the development of hydrophilic organic monoliths with reduced non-specific interactions and high-protein grafting capacity remains a hot topic. In this work, we propose the one-step synthesis of a diol organic monolith to replace the gold-standard epoxy-based organic monoliths (which require post-modification, namely hydrolysis, prior to use). The synthesis of this new monolith builds upon the use of N-N’-Methylenebis(acrylamide) (MBA), as a hydrophilic crosslinker, and 2,3-dihydroxypropyl methacrylate (DHPMA), a diol monomer that eliminates the time-consuming epoxy ring opening step and its associated side reactions. The optimization of one-step synthesis parameters led to a monolith with a satisfactory permeability ((4.8 ± 0.5) × 10−14 m2), high efficiency (117,600 plates/m at optimum flow velocity (uopt = 0.09 cm s−1)) and reduced non-specific interactions. It is exemplified by its separation ability in the HILIC mode (separation of nucleosides), and by the retention data set of 41 test solutes, which were used to evaluate the non-specific interactions. This new poly(DHPMA-co-MBA) monolith has not only hydrophilic surface properties, but also improved protein grafting capacity compared to the glycidyl-based monolith (13 ± 0.7 pmol cm−1). The potential of this monolith is illustrated in affinity chromatography, where the concanavalin ligands are ranked according to their Kd values.
Full article
(This article belongs to the Section Chromatographic Separations)
►▼
Show Figures
Graphical abstract
Open AccessArticle
Chemometric Approach of Different Extraction Conditions on Scavenging Activity of Helichrisym italicum (Roth) G. Don Extracts
by
, , , , , , and
Separations 2023, 10(8), 436; https://doi.org/10.3390/separations10080436 - 02 Aug 2023
Abstract
Helichrysum italicum (Roth) G. Don is widely used as a liver stimulant and diuretic, as well as an antioxidant, antiallergenic, antimicrobial, antiviral, choleric, and antihypertensive agent. Therefore, the purpose of this study was to analyze the different extracts of H. italicum and to
[...] Read more.
Helichrysum italicum (Roth) G. Don is widely used as a liver stimulant and diuretic, as well as an antioxidant, antiallergenic, antimicrobial, antiviral, choleric, and antihypertensive agent. Therefore, the purpose of this study was to analyze the different extracts of H. italicum and to determine the relationship between total phenolic/flavonoid content and antioxidant activity. Plant material was pulverized to be 0.3 and 2 mm, and the extraction was obtained with 5 mL of different ethanol concentrations (30, 40, 50, 60, 96%, v/v) and water during different periods (10, 30, 60 min and 24 h) and with concentrated methanol (95%, v/v), dichloromethane, acetone, chloroform, ethyl acetate, and hexane during 10 and 30 min due to solvents volatility. Extracts were evaporated on the rotary evaporator. Total phenolic and flavonoid content were determined via spectrophotometric measurement, and antioxidant capacity was investigated by the inhibitory activity of DPPH radicals. The group of 92 extracts described through their TPC, flavonoids, yield, and IC50 values were subjected to PCA and HCA analysis. PCA and HCA were performed using Statistica v 10.0 software. The number of phenolic compounds ranged from 2.58 ± 0.1 to 43.60 ± 0.3 mg GAE/g of dry extract, and total flavonoids content ranged from 0.25 ± 0.01 to 23.96 ± 0.19 mg QE/g of dry extract. All extracts showed good antioxidant activity with an IC50 value in the range from 4.92 ± 0.2 to 231.12 ± 1.9 μg/mL. PCA and HCA analysis were performed on the whole data set comprising 92 extracts for both particle sizes (s0.3 and s2) as well as heat map analysis. It was found that the increased time of extraction, concentration of ethanol, and degree of fragmentation enhances the quality of the extracts in terms of phenolic component content and antioxidant effects. Applied chemometric analysis contributed to the extraction conditions grouping, overview, and selection in order to distinguish the extracts with the promising TPC, flavonoids, yield, and IC50 values.
Full article
(This article belongs to the Special Issue Extraction and Analysis of Active Ingredients from Natural Products)
►▼
Show Figures
Graphical abstract
Open AccessArticle
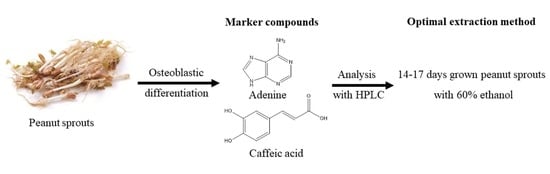
Isolation of Osteoblastic Differentiation-Inducing Constituents from Peanut Sprouts and Development of Optimal Extraction Method
Separations 2023, 10(8), 435; https://doi.org/10.3390/separations10080435 - 01 Aug 2023
Abstract
Osteoporosis, one of the most common bone diseases, results from an imbalance between bone formation and resorption. Osteoblasts are primarily involved in bone formation, whereas osteoclasts are involved in bone resorption. Therefore, any substances that can increase osteoblastic differentiation would be beneficial for
[...] Read more.
Osteoporosis, one of the most common bone diseases, results from an imbalance between bone formation and resorption. Osteoblasts are primarily involved in bone formation, whereas osteoclasts are involved in bone resorption. Therefore, any substances that can increase osteoblastic differentiation would be beneficial for the prevention or treatment of osteoporosis. In this study, peanut sprouts, a germinated product of peanuts, significantly enhanced osteoblastic differentiation of pre-osteoblastic MC3T3-E1 cells, as determined by the alkaline phosphatase (ALP) assay. The ethyl acetate fraction of peanut sprout extract was subjected to diverse column chromatographies using silica gel, ODS, and Sephadex LH-20 as stationary phases, and three nucleobases, namely, adenine, uracil, and thymine, and three phenolic acids, including caffeic acid, coumaric acid, and ferulic acid, were isolated as active constituents. In particular, adenine at 4 μg/mL and caffeic acid at 1 μg/mL increased ALP activity by 40 and 30%, respectively, compared with the osteoblastic differentiation medium-treated group, and these two compounds were set as marker compounds. Furthermore, extraction of peanut sprouts grown for 14–17 days with 60% ethanol was the best condition to obtain a high quantity of extract for peanut sprouts by analyzing the contents of marker compounds through HPLC. Together, these results suggest that peanut sprouts and their isolated compounds significantly enhance osteoblastic differentiation. Therefore, peanut sprouts have the potential to be developed as functional foods against osteoporosis.
Full article
(This article belongs to the Special Issue Separation, Analysis and Biological Evaluation of Bioactive Compounds)
►▼
Show Figures
Graphical abstract
Open AccessArticle
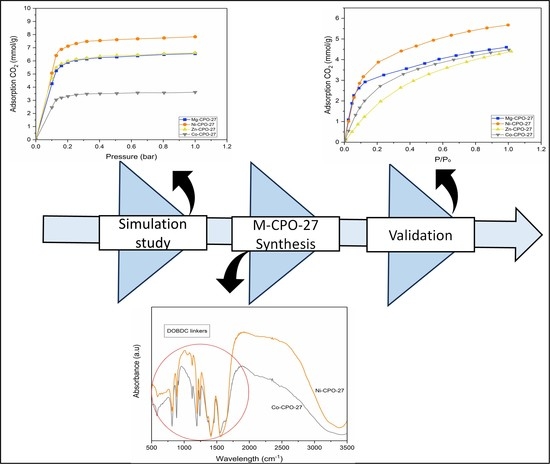
Screening and Experimental Validation for Selection of Open Metal Sites Metal-Organic Framework (M-CPO-27, M = Co, Mg, Ni and Zn) to Capture CO2
by
, , , , and
Separations 2023, 10(8), 434; https://doi.org/10.3390/separations10080434 - 01 Aug 2023
Abstract
The release of CO2 into the atmosphere has become a primary issue nowadays. Recently, researchers found Metal-Organic Frameworks M-CPO-27 (M = Mg, Co, Ni, and Zn) to be revolutionary for CO2 adsorption due to the presence of open metal sites enhancing
[...] Read more.
The release of CO2 into the atmosphere has become a primary issue nowadays. Recently, researchers found Metal-Organic Frameworks M-CPO-27 (M = Mg, Co, Ni, and Zn) to be revolutionary for CO2 adsorption due to the presence of open metal sites enhancing CO2 binding and leading to higher capacity. This study aims to select the best metal center for CPO-27 with the high performance of CO2 adsorption by screening metal centers using simulation as a preliminary selection method. Then, the different metal centers were synthesized using the solvothermal process for validation. The synthesis of MOFs is confirmed through PXRD and FTIR analysis. Subsequently, by using simulation and experimental methods, it is discovered that Ni-CPO-27 gives the best performance compared with magnesium, zinc, and cobalt metal centers. The CO2 adsorption capacity of synthesized Ni-CPO-27 is 5.6 mmol/g, which is almost 20% higher than other MOFs. In conclusion, the prospective outcome of changing the metal from Mg-CPO-27 to Ni-CPO-27 would be advantageous in this investigation owing to its excellent performance in capturing CO2.
Full article
(This article belongs to the Special Issue Advanced Techniques in the Separation Processes and Development of Novel Adsorbents)
►▼
Show Figures
Graphical abstract
Open AccessArticle
Water-Based Microwave-Assisted Extraction of Pigments from Madder Optimized by a Box–Behnken Design
Separations 2023, 10(8), 433; https://doi.org/10.3390/separations10080433 - 30 Jul 2023
Abstract
Water is one of the greenest and most accessible solvents. To harness its potential, the water-based microwave-assisted extraction (MAE) of pigments from madder was optimized by the response surface methodology (RSM) using a Box–Behnken experimental design. The extract that presented both the highest
[...] Read more.
Water is one of the greenest and most accessible solvents. To harness its potential, the water-based microwave-assisted extraction (MAE) of pigments from madder was optimized by the response surface methodology (RSM) using a Box–Behnken experimental design. The extract that presented both the highest extraction yield and the most intense color was obtained after a 30-s cycle at 1000 W using 100 mg of madder for 20 mL of water. This water-based MAE was more efficient than Soxhlet extraction and proved comparable to hydroalcoholic MAE. The optimized extract was further characterized using UHPLC-HRMS/MS to identify its main compounds. Glycosylated flavonoids and anthraquinones were putatively identified, as well as free anthraquinones, generally found in madder. The microwave-assisted extraction extended the range of polarity of the extracted compounds, making the water more versatile.
Full article
(This article belongs to the Special Issue Separation, Extraction and Purification of Natural Products)
►▼
Show Figures
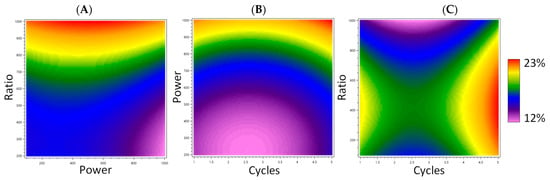
Figure 1
Open AccessArticle
Study of Estimated Ultimate Recovery Prediction and Multi-Stage Supercharging Technology for Shale Gas Wells
Separations 2023, 10(8), 432; https://doi.org/10.3390/separations10080432 - 29 Jul 2023
Abstract
The development of shale gas reservoirs often involves the utilization of horizontal well segmental multi-stage fracturing techniques. However, these reservoirs face challenges, such as rapid initial wellhead pressure and production decline, leading to extended periods of low-pressure production. To address these issues and
[...] Read more.
The development of shale gas reservoirs often involves the utilization of horizontal well segmental multi-stage fracturing techniques. However, these reservoirs face challenges, such as rapid initial wellhead pressure and production decline, leading to extended periods of low-pressure production. To address these issues and enhance the production during the low-pressure stage, pressurized mining is considered as an effective measure. Determining the appropriate pressurization target and method for the shale gas wells is of great practical significance for ensuring stable production in shale gas fields. This study takes into account the current development status of shale gas fields and proposes a three-stage pressurization process. The process involves primary supercharging at the center station of the block, secondary supercharging at the gas collecting station, and the introduction of a small booster device located behind the platform separator and in front of the outbound valve group. By incorporating a compressor, the wellhead pressure can be reduced to 0.4 MPa, resulting in a daily output of 12,000 to 14,000 cubic meters from the platform. Using a critical liquid-carrying model for shale gas horizontal wells, this study demonstrates that reducing the wellhead pressure decreases the critical flow of liquid, thereby facilitating the discharge of the accumulated fluid from the gas well. Additionally, the formation pressure of shale gas wells is estimated using the mass balance method. This study calculates the cumulative production of different IPR curves based on the formation pressure. It develops a dynamic production decline model for gas outlet wells and establishes a relationship between the pressure depletion of gas reservoirs and the cumulative gas production before and after pressurization of H10 −2 and H10 −3 wells. The final estimated ultimate recovery of two wells is calculated. In conclusion, the implementation of multi-stage pressurization, as proposed in this study, effectively enhances the production of, and holds practical significance for, stable development of shale gas fields.
Full article
(This article belongs to the Topic Oil, Gas and Water Separation Research)
►▼
Show Figures
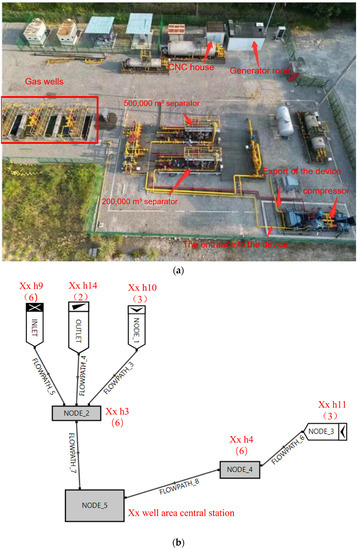
Figure 1
Open AccessArticle
Separation of (2E,4E)-deca-2,4-dienal from a Biocatalytic Reaction Mixture Using Hydrophobic Adsorbents
by
, , , , and
Separations 2023, 10(8), 431; https://doi.org/10.3390/separations10080431 - 29 Jul 2023
Abstract
Biocatalytic oxidation of a fatty acid preparation by enzymes released from a plant extract provided (2E,4E)-deca-2,4-dienal (2,4-DDAL), a valuable natural aroma. The post-reaction mixture contained several other compounds, from which 2,4-DDAL must be recovered. These included the main by-product trans-2-hexenal (2-HAL) and the
[...] Read more.
Biocatalytic oxidation of a fatty acid preparation by enzymes released from a plant extract provided (2E,4E)-deca-2,4-dienal (2,4-DDAL), a valuable natural aroma. The post-reaction mixture contained several other compounds, from which 2,4-DDAL must be recovered. These included the main by-product trans-2-hexenal (2-HAL) and the unreacted substrate composed mainly of linoleic, palmitic, and oleic acids. The adsorption capacity and selectivity of 17 hydrophobic adsorbents were examined in batch equilibrium experiments. The highest binding capacity for 2,4-DDAL was around 30 mg/g. Due to a large excess of fatty acids in the mixture, the total adsorbed amounts of other components analyzed reached the values up to 150 mg/g. In addition, 2-HAL was always adsorbed more strongly than 2,4-DDAL, whereas fatty acids were mostly not adsorbed as effectively. The best-performing adsorbent, AmberLite FPX66, had a selectivity ratio of 6 for 2,4-DDAL with respect to the key impurity and linoleic acid. Ethanol, isopropanol, and acetone were examined as potential desorbents using batch adsorption experiments on AmberLite FPX66 from 2,4-DDAL solutions in organic solvents. Ethanol was found to be the best choice. Based on the batch equilibrium data, a column elution experiment was performed to purify 2,4-DDAL from the biocatalytic reaction mixture using elution with 96% ethanol.
Full article
(This article belongs to the Collection Feature Paper Collection in Section 'Purification Technology')
►▼
Show Figures
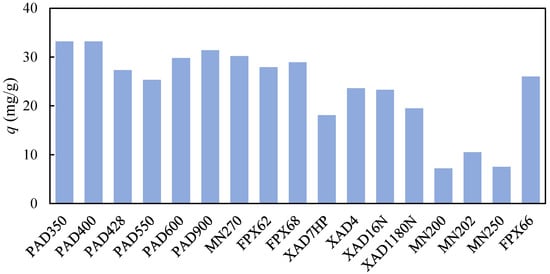
Figure 1
Open AccessArticle
Superior Removal of Toxic Cr(VI) from Wastewaters by Natural Pine Bark
Separations 2023, 10(8), 430; https://doi.org/10.3390/separations10080430 - 28 Jul 2023
Abstract
Hexavalent chromium (Cr(VI)) is one of the most toxic heavy metals found in industrial wastewater, so many researchers are working to develop efficient and environmentally friendly removal methods. It has been reported that natural biomass and its derivatives can be used to treat
[...] Read more.
Hexavalent chromium (Cr(VI)) is one of the most toxic heavy metals found in industrial wastewater, so many researchers are working to develop efficient and environmentally friendly removal methods. It has been reported that natural biomass and its derivatives can be used to treat wastewaters containing Cr(VI). However, biomass with sufficient Cr(VI) removal performance to replace the existing chemical method, which is cheap and simple, has not been reported yet. This study reports that inexpensive, abundant, and commercially available pine bark has the highest Cr(VI) removal capacity (i.e., 376.3 mg/g) compared to biomass reported elsewhere. This value is six times higher than the theoretical value of an inorganic reducing agent (iron(II) sulfate heptahydrate). The main mechanism of Cr(VI) removal by pine bark was clearly identified through kinetic experiments, Fourier-transform infrared spectrometer, and X-ray photoelectron spectroscopy analyses, which were used to study the compositions, functional groups, and bonding states of pine bark. It was found that pine bark consists of various acidic functional groups that can act as electron donors to promote the removal of Cr(VI) through redox reactions. In conclusion, pine bark may be a promising candidate for the removal of Cr(VI) from wastewater, owing to its excellent removal capacity.
Full article
(This article belongs to the Special Issue Removal of Metals or Other Toxic Substances from Wastewaters by Natural Sorbents)
►▼
Show Figures
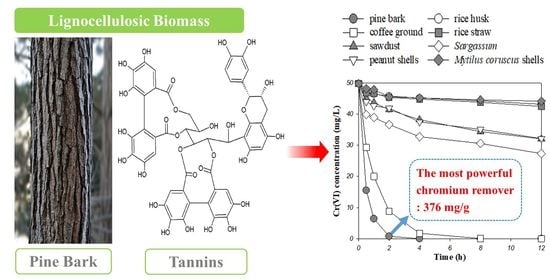
Graphical abstract
Open AccessArticle
Determination of β2-Agonist Residues in Meat Samples by Gas Chromatography-Mass Spectrometry with N-Doped Carbon Dots in Molecular Sieves
Separations 2023, 10(8), 429; https://doi.org/10.3390/separations10080429 - 28 Jul 2023
Abstract
►▼
Show Figures
A simple, effective, and highly sensitive analytical approach was created and applied in this study for the accurate measurement of three β2-agonist residues (clenbuterol, salbutamol, and ractopamine) in meat samples. In the course of the experiment, new adsorbent molecular sieves (ZMS)@nitrogen-doped
[...] Read more.
A simple, effective, and highly sensitive analytical approach was created and applied in this study for the accurate measurement of three β2-agonist residues (clenbuterol, salbutamol, and ractopamine) in meat samples. In the course of the experiment, new adsorbent molecular sieves (ZMS)@nitrogen-doped carbon quantum dots (N-CQDs) composite materials were synthesized with the aid of hydrothermal synthesis. The composite adsorbent materials were prepared and characterized through scanning electron microscopy, transmission electron microscope, X-ray photoelectron spectroscopy, fluorescence, and zeta potential. Four determinants affecting the extraction and elution’s efficiency, such as the amount of adsorbent, the extraction time, desorption time, and the amount of extraction salt, were substantially optimized. The analytes were quantified by gas chromatography–mass spectrometry. Final results of the methodological validation reflected that the [email protected] composite materials were able to adsorb three β2-agonist residues well and had good reproducibility. In the meantime, all analytes indicated good linearity with coefficient of determination R2 ≥ 0.9908. The limit of detection was 0.7–2.0 ng·g−1, the limit of quantification varied from 2.4 to 5.0 ng·g−1, the precision was lower than 11.9%, and the spiked recoveries were in the range of 79.5–97.8%. To sum up, the proposed approach was quite effective, reliable, and convenient for the simultaneous analysis of multiple β2-agonist residues. Consequently, this kind of approach was successfully applied for the analysis of such compounds in meat samples.
Full article

Figure 1
Open AccessCommunication
Isolation and Structural Elucidation of Unreported Prenylhydroquinone Glycoside from Sedum kamtschaticum Leaves and Its Effect on Hyperphosphorylated Tau Production in Aβ1–42-Treated SH-SY5Y Cells
by
, , , , , and
Separations 2023, 10(8), 428; https://doi.org/10.3390/separations10080428 - 28 Jul 2023
Abstract
Sedum kamtschaticum Fischer, of the Crassulaceae family, is a perennial and medicinal plant used in Asian folk medicine to alleviate inflammatory disease and improve blood circulation. As part of our ongoing exploration into natural products, seeking to identifying bioactive compounds, we characterized, identified,
[...] Read more.
Sedum kamtschaticum Fischer, of the Crassulaceae family, is a perennial and medicinal plant used in Asian folk medicine to alleviate inflammatory disease and improve blood circulation. As part of our ongoing exploration into natural products, seeking to identifying bioactive compounds, we characterized, identified, and isolated an unreported bioactive compound, prenylhydroquinone glycoside (1), which we named kirinchoside from S. kamtschaticum leaves. Using high-resolution (HR)-ESIMS, NMR spectroscopic data, and enzymatic hydrolysis, followed by LC–MS analysis, we determined the structure of this isolated compound. Despite a previous report on the planar structure of compound 1 (kirinchoside), the absolute configuration of 1 had not been verified. We investigated the effects of kirinchoside on hyperphosphorylated tau (p-tau) accumulation, a hallmark of Alzheimer’s disease (AD) progression. We observed that treatment with 5 μM kirinchoside suppressed p-tau levels by 16.9% in amyloid β (Aβ)1–42-treated SH-SY5Y cells, compared to the negative control. These findings indicate that kirinchoside, an unreported prenylhydroquinone glycoside found in S. kamtschaticum leaves, could be a candidate preventive agent against AD via inhibition of p-tau accumulation.
Full article
(This article belongs to the Collection Feature Paper Collection in Section 'Purification Technology')
►▼
Show Figures
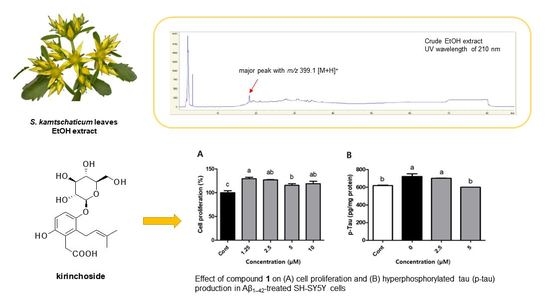
Graphical abstract
Open AccessArticle
Biosynthesis of Salbutamol-4′-O-sulfate as Reference for Identification of Intake Routes and Enantiopure Salbutamol Administration by Achiral UHPLC-MS/MS
by
, , , , , , , and
Separations 2023, 10(8), 427; https://doi.org/10.3390/separations10080427 - 28 Jul 2023
Abstract
The aim of the study was a comprehensive and quantitative determination of salbutamol and its sulfoconjugated major metabolite in urine samples using achiral ultrahigh performance liquid chromatography-tandem mass spectrometry (UHPLC-MS/MS). Therefore, salbutamol-4′-O-sulfate was biosynthesized as a reference using genetically modified fission
[...] Read more.
The aim of the study was a comprehensive and quantitative determination of salbutamol and its sulfoconjugated major metabolite in urine samples using achiral ultrahigh performance liquid chromatography-tandem mass spectrometry (UHPLC-MS/MS). Therefore, salbutamol-4′-O-sulfate was biosynthesized as a reference using genetically modified fission yeast cells, and the product was subsequently characterized by NMR and HRMS. In competitive sports, salbutamol is classified as a prohibited drug; however, inhalation at therapeutic doses is permitted with a maximum allowance of 600 µg/8 h. In contrast, the enantiopure levosalbutamol is prohibited under any condition. For analytical discrimination, the amount of salbutamol and its main metabolite excreted in the urine was studied. As proof of concept, a longitudinal study in one healthy volunteer was performed in order to investigate excreted amounts and to study potential discrimination using achiral chromatography. Discrimination of administration of racemic salbutamol or the enantiopure levosalbutamol was not achieved by solely analyzing salbutamol as the parent compound. However, a distinction was possible by evaluation of the proportion of salbutamol-4′-O-sulfate in relation to salbutamol. Therefore, reference material of metabolites is of great importance in doping control, especially for threshold substances.
Full article
(This article belongs to the Special Issue Application of Hyphenated Techniques in Bioanalysis and Metabolite Identification)
►▼
Show Figures
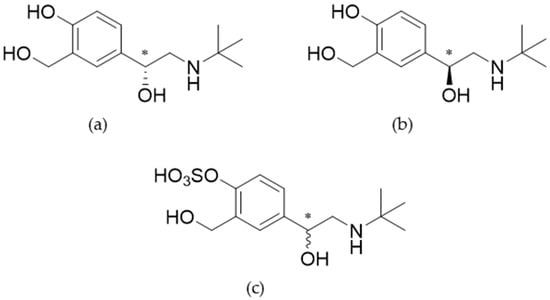
Figure 1
Open AccessArticle
Removal of Azo Dyes Orange II and Reactive Black 5 from Aqueous Solutions by Adsorption on Chitosan Beads Modified with Choline Chloride: Urea Deep Eutectic Solvent and FeO
Separations 2023, 10(8), 426; https://doi.org/10.3390/separations10080426 - 27 Jul 2023
Abstract
The removal of the azo dyes Orange II (OII) and Reactive Black 5 (RB5) from aqueous solutions was studied using three types of adsorbents derived from chitosan: unmodified chitosan beads (un-Ch), chitosan beads tuned with a deep eutectic solvent (DES) composed of choline
[...] Read more.
The removal of the azo dyes Orange II (OII) and Reactive Black 5 (RB5) from aqueous solutions was studied using three types of adsorbents derived from chitosan: unmodified chitosan beads (un-Ch), chitosan beads tuned with a deep eutectic solvent (DES) composed of choline chloride:urea at a 1:2 molar ratio (Ch-DES), along with FeO nanoparticles added to chitosan and modified with DES (Ch-FeO-DES). Both dyes were effectively removed in 3–4 h when Ch-DES beads were used, at a dye concentration range of 25–50 mg/L. The modification with DES improved the removal efficiency to achieve increases of around 32% for OII and 17% for RB5. The adsorbent dosage and the initial dye concentration influenced the adsorption process. Isotherm adsorption models (Langmuir, Freundlich, and Temkin) and kinetic models (pseudo-first-order, pseudo-second-order, intraparticle diffusion, and Elovich models) were applied to identify the adsorption behavior. The process could be characterized by employing the pseudo-second-order kinetic model and fitting it to the monolayer Langmuir isotherm. The adsorption occurred on a monolayer with no interaction among dye molecules when electrostatic forces would determine the attaching of dye molecules on the adsorbent. OII was efficiently desorbed by an alkaline solution, while RB5 was more strongly attached. The adsorbent could be recycled, after regeneration with an acid solution, for at least 5 reuse cycles while still achieving effective OII removal. Reactivation with a diluted EDTA solution allowed the authors to recycle RB5-attached beads, achieving optimum removal in two further uses.
Full article
(This article belongs to the Special Issue Feature Papers in Separations from Editorial Board Members)
►▼
Show Figures

Figure 1
Open AccessArticle
Effects of Dissolved Organic Matter on the Release of Soluble Phosphorus and Fluoride Ion from Phosphate Ore
Separations 2023, 10(8), 425; https://doi.org/10.3390/separations10080425 - 27 Jul 2023
Abstract
Unreasonable storage of phosphate ore is becoming an important pathway causing phosphate pollution in the surrounding aquatic environment. However, there is little research on the influence of dissolved organic matter (DOM) in water on the fate of phosphate ore. Here, we collected phosphate
[...] Read more.
Unreasonable storage of phosphate ore is becoming an important pathway causing phosphate pollution in the surrounding aquatic environment. However, there is little research on the influence of dissolved organic matter (DOM) in water on the fate of phosphate ore. Here, we collected phosphate ores from two phosphate mines along the coast of Tanglang River and studied the effects of DOM concentrations and pH on the release of soluble active phosphorus (SRP) and fluoride ion (F−) from phosphate ores using humic acid (HA) as the representative of DOM. Based on the analysis of ZP, FTIR, XPS, and SEM, the influence mechanism of HA was revealed. The results showed that HA efficiently promoted the release of SRP and F− from phosphate ore. With decreasing pH, the P release increased in both water and HA solutions in general. The beneficial influence of HA on the release of SRP and F− from phosphate ore was ascribed to the introduction of oxygen-containing functional groups by HA, which altered the surface properties and enhanced the dispersion stability of phosphate ore. These findings provided new insights into the dispersion behavior of phosphate ore, which is helpful in promoting the pollution control and management strategy of phosphate ore.
Full article
(This article belongs to the Section Environmental Separations)
►▼
Show Figures

Figure 1
Open AccessReview
Commercial Anion Exchange Membranes (AEMs) for Fuel Cell and Water Electrolyzer Applications: Performance, Durability, and Materials Advancement
Separations 2023, 10(8), 424; https://doi.org/10.3390/separations10080424 - 26 Jul 2023
Abstract
The utilization of anion exchange membranes (AEMs) has revolutionized the field of electrochemical applications, particularly in water electrolysis and fuel cells. This review paper provides a comprehensive analysis of recent studies conducted on various commercial AEMs, including FAA3-50, Sustainion, Aemion™, XION Composite, and
[...] Read more.
The utilization of anion exchange membranes (AEMs) has revolutionized the field of electrochemical applications, particularly in water electrolysis and fuel cells. This review paper provides a comprehensive analysis of recent studies conducted on various commercial AEMs, including FAA3-50, Sustainion, Aemion™, XION Composite, and PiperION™ membranes, with a focus on their performance and durability in AEM water electrolysis (AEMWE) and AEM fuel cells (AEMFCs). The discussed studies highlight the exceptional potential of these membranes in achieving high current densities, stable operation, and extended durability. Furthermore, the integration of innovative catalysts, such as nitrogen-doped graphene and Raney nickel, has demonstrated significant improvements in performance. Additionally, the exploration of PGM-free catalysts, such as Ag/C, for AEMFC cathodes has unveiled promising prospects for cost-effective and sustainable fuel cell systems. Future research directions are identified, encompassing the optimization of membrane properties, investigation of alternative catalyst materials, and assessment of performance under diverse operating conditions. The findings underscore the versatility and suitability of these commercial AEMs in water electrolysis and fuel cell applications, paving the way for the advancement of efficient and environmentally benign energy technologies. This review paper serves as a valuable resource for researchers, engineers, and industry professionals seeking to enhance the performance and durability of AEMs in various electrochemical applications.
Full article
(This article belongs to the Special Issue Fuel Cell Membranes and Adsorption Technologies)
►▼
Show Figures

Figure 1

Journal Menu
► ▼ Journal Menu-
- Separations Home
- Aims & Scope
- Editorial Board
- Reviewer Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Society Collaborations
- Conferences
- Editorial Office
Journal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Analytica, Molecules, Nanomaterials, Polymers, Separations
Nanomaterials in Green Analytical Chemistry
Topic Editors: George Zachariadis, Rosa Peñalver, Natalia ManousiDeadline: 15 August 2023
Topic in
Energies, Polymers, Remote Sensing, Separations, Water
Oil, Gas and Water Separation Research
Topic Editors: Liming Zhang, Kai ZhangDeadline: 15 September 2023
Topic in
Energies, Membranes, Molecules, Separations, Water
Sustainable Water Purification Technologies for Multiple Applications
Topic Editors: Marco Pellegrini, Cesare Saccani, Alessandro GuzziniDeadline: 15 October 2023
Topic in
Analytica, Antioxidants, Applied Sciences, Molecules, Separations
New Analytical Methods in Plant Active Components Analysis
Topic Editors: Filomena Lelario, Giuliana Bianco, Radosław KowalskiDeadline: 31 October 2023

Conferences
Special Issues
Special Issue in
Separations
Porous Materials for Separation of Gas and Chemicals
Guest Editor: Xin ZhangDeadline: 10 August 2023
Special Issue in
Separations
Determination of Pesticides and Drug Residues by Liquid Chromatography-Mass Spectrometry
Guest Editor: Andreia FreitasDeadline: 25 August 2023
Special Issue in
Separations
Research on Coupling of Electrochemical-Membrane Separation
Guest Editor: Wen ZhangDeadline: 31 August 2023
Special Issue in
Separations
Food Volatilomics: Separation and Extraction of Food Aromatic Compounds
Guest Editor: Zipora TietelDeadline: 20 September 2023
Topical Collections
Topical Collection in
Separations
Feature Paper Collection in Section 'Materials in Separation Science'
Collection Editors: Dimosthenis Giokas, Manolis Manos
Topical Collection in
Separations
Recent Trends in the Separation of Natural Products and Pharmaceuticals
Collection Editors: Paraskevas D. Tzanavaras, Susanne Wiedmer
Topical Collection in
Separations
Synthetic Membrane Separation Science and Technology
Collection Editors: Mohamed Khayet, Elena Guillen Burrieza
Topical Collection in
Separations
State of the Art in Separation Science
Collection Editor: Victoria Samanidou















