Journal Description
Organics
Organics
is an international, peer-reviewed, open access journal on organic chemistry published quarterly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within CAPlus / SciFinder, and other databases.
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 20.6 days after submission; acceptance to publication is undertaken in 6.2 days (median values for papers published in this journal in the first half of 2023).
- Recognition of Reviewers: APC discount vouchers, optional signed peer review, and reviewer names published annually in the journal.
Latest Articles
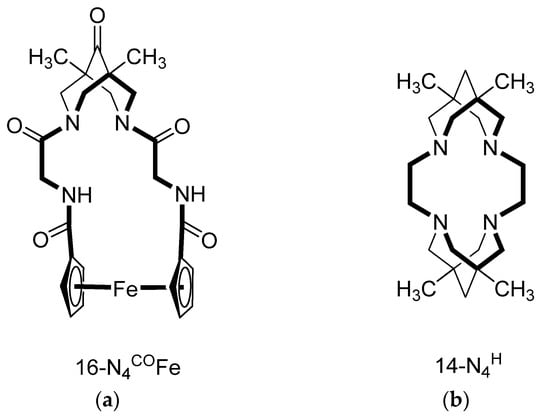
Bispidine-Based Macrocycles: Achievements and Perspectives
Organics 2023, 4(3), 417-437; https://doi.org/10.3390/org4030030 - 24 Jul 2023
Abstract
►
Show Figures
This review presents all currently known macroheterocyclic compounds that include a bispidine (3,7-diazabicyclo[3.3.1]nonane) fragment in their structure. A classification of bispidine-containing macroheterocycles, which is based on the ring size and the nature of bispidinic nitrogen atoms, is suggested. Synthetic approaches to the studied
[...] Read more.
This review presents all currently known macroheterocyclic compounds that include a bispidine (3,7-diazabicyclo[3.3.1]nonane) fragment in their structure. A classification of bispidine-containing macroheterocycles, which is based on the ring size and the nature of bispidinic nitrogen atoms, is suggested. Synthetic approaches to the studied compounds are classified and considered. The features of the crystal structures and solution behavior of bispidine macroheterocycles are analyzed. Prospects for the development of these organic receptors are proposed.
Full article
Open AccessArticle
Synthesis of an Electrodeficient Dipyridylbenzene-like Terdentate Ligand: Cyclometallating Ligand for Highly Emitting Iridium(III) and Platinum(II) Complexes
Organics 2023, 4(3), 403-416; https://doi.org/10.3390/org4030029 - 14 Jul 2023
Abstract
Cyclometallated iridium(III) and platinum(II) complexes are intensely used in optoelectronics for their photophysical properties and ability to convert excitons from singlet to triplet state, thus improving the device efficiency. In this contribution, we report the multi-steps synthesis of an electrodeficient dipyridylbenzene-like terdentate ligand
[...] Read more.
Cyclometallated iridium(III) and platinum(II) complexes are intensely used in optoelectronics for their photophysical properties and ability to convert excitons from singlet to triplet state, thus improving the device efficiency. In this contribution, we report the multi-steps synthesis of an electrodeficient dipyridylbenzene-like terdentate ligand [N^C^N], namely 2′,6′-dimethyl-2,3′:5′,2″-terpyridine (6), with 18% overall yield. Compound 6 has been employed to synthesize two phosphorescent complexes of platinum(II) and iridium(III), namely compounds 7 and 8, respectively. Both complexes have been characterized by NMR and high resolution mass spectrometry, and demonstrate high luminescence quantum yields in a deaerated solution at room temperature, with 18% and 61% for 7 and 8, respectively. If the iridium(III) complex displays similar emission properties to [Ir(dpyx)(ppy)Cl] (dpyx = 3,5-dimethyl-2,6-dipyridylbenzene and ppy = 2- phenylpyridine), the platinum(II) derivative, with λem = 470 nm, is a rare example of a fluorine atom-free blue emitting [N^C^N]PtCl complex.
Full article
(This article belongs to the Collection Advanced Research Papers in Organics)
►▼
Show Figures
Figure 1
Open AccessReview
An Overview of Some Reactive Routes to Flame-Retardant Fibre-Forming Polymers: Polypropylene and Polyacrylonitrile
Organics 2023, 4(3), 386-402; https://doi.org/10.3390/org4030028 - 12 Jul 2023
Abstract
The thermal degradation and flammability characteristics of some common fibre-forming polymers, such as polypropylene (PP) and polyacrylonitrile (PAN), are described in this review paper. The flame retardance of these polymers is principally affected by reactive routes that were primarily developed in our laboratories.
[...] Read more.
The thermal degradation and flammability characteristics of some common fibre-forming polymers, such as polypropylene (PP) and polyacrylonitrile (PAN), are described in this review paper. The flame retardance of these polymers is principally affected by reactive routes that were primarily developed in our laboratories. The modifying groups that are incorporated into polymeric chains include phosphorus- or phosphorus/nitrogen-containing moieties in different chemical environments. The degradation characteristics and extent of flame retardance were mainly evaluated using routine thermal and calorimetric techniques. Elements of flame-retardant mechanisms occurring in the condensed and vapour phases were also identified. Furthermore, we also explored the effects of molecularly dispersed β-cyclodextrin, including its physical mixtures, on the thermal and combustion characteristics of PAN. Given that both types of polymers are often used in the form of fibres, and that the aspect ratio of fibrous materials is relatively high, even nominal enhancements in their fire retardance are highly welcomed. Hence, the preliminary results of our research on chemically modified PAN incorporating molecularly dispersed β-cyclodextrin are encouraging in terms of their enhanced fire retardance, and hence this field warrants further exploration.
Full article
(This article belongs to the Special Issue Progress in Synthesis and Applications of Phosphorus-Containing Compounds)
►▼
Show Figures
Figure 1
Open AccessArticle
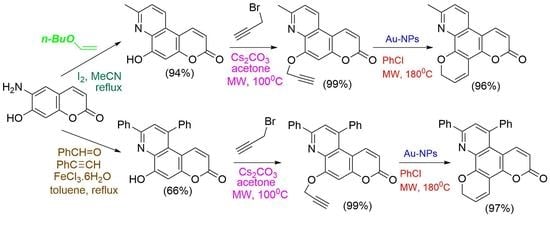
Synthesis and Biological Evaluation of Substituted Fused Dipyranoquinolinones
by
, , and
Organics 2023, 4(3), 364-385; https://doi.org/10.3390/org4030027 - 10 Jul 2023
Abstract
New methyl-substituted, and diphenyl-substituted fused dipyranoquinolinones are prepared in excellent yields via the triple bond activation and 6-endo-dig cyclization of propargyloxycoumarin derivatives by gold nanoparticles supported on TiO2 in chlorobenzene under microwave irradiation. In the absence of gold nanoparticles, the methyl-substituted propargyloxycoumarin
[...] Read more.
New methyl-substituted, and diphenyl-substituted fused dipyranoquinolinones are prepared in excellent yields via the triple bond activation and 6-endo-dig cyclization of propargyloxycoumarin derivatives by gold nanoparticles supported on TiO2 in chlorobenzene under microwave irradiation. In the absence of gold nanoparticles, the methyl-substituted propargyloxycoumarin derivatives resulted in fused furopyranoquinolinones through Claisen rearrangement and 5-exo-dig cyclization. The intermediate propargyloxy-fused pyridocoumarins are prepared by propargylation of the corresponding hydroxy-fused pyridocoumarins. The methyl-substituted derivatives of the latter are synthesized in excellent yield by the three-component reaction of amino hydroxycoumarin with n-butyl vinyl ether under iodine catalysis. The diphenyl-substituted derivatives of hydroxy-fused pyridocoumarins are obtained, also, by the three-component reaction of amino hydroxycoumarin with benzaldehyde and phenyl acetylene catalyzed by iron (III) chloride. Preliminary biological tests of the title compounds indicated lipoxygenase (LOX) (EC 1.13.11.12) inhibitory activity (60–100 μM), whereas compound 28a, with IC50 = 10 μM, was found to be a potent LOX inhibitor and a possible lead compound. Only compounds 10b and 28b significantly inhibited lipid peroxidation.
Full article
(This article belongs to the Collection Advanced Research Papers in Organics)
►▼
Show Figures
Graphical abstract
Open AccessReview
Indole-Based Macrocyclization by Metal-Catalyzed Approaches
Organics 2023, 4(3), 333-363; https://doi.org/10.3390/org4030026 - 04 Jul 2023
Abstract
This review is dedicated to the different varieties of macrocycles synthesis bearing indole units in their architecture by metal-catalyzed strategies. The progress of the new macrocyclization approaches is persisted be a keen area of research. Macrocycles contain a wide variety of molecules, and
[...] Read more.
This review is dedicated to the different varieties of macrocycles synthesis bearing indole units in their architecture by metal-catalyzed strategies. The progress of the new macrocyclization approaches is persisted be a keen area of research. Macrocycles contain a wide variety of molecules, and among those, heteroaryl motifs are valuable constituents that provide an attractive feature to macrocyclic systems. Indole represents one of the privileged pharmacophores against a variety of targets with various biological applications. Among the nitrogen-based heterocycles, indole plays a prominent role in organic synthesis, medicinal chemistry, pharmaceuticals, natural products synthesis, agrochemicals, dye and fragrances, and drug design. These scaffolds are widely distributed in several bioactive natural products and synthetic macrocycles constructed against a specific biochemical target and the most common constituents of naturally occurring molecules. Due to its immense importance, the progress of novel approaches for the synthesis of indole-based scaffolds has increased steadily. The majority of the macrocycles synthesis proceeds through the macrolactamization and macrolactonization, as well as the C–C bond macrocyclization process described by metal-catalyzed ring-closing metathesis (RCM) and coupling reactions. Among macrocyclizations, metal-catalyzed approaches are considered one of the most powerful tools for synthetic chemists in the design of a variety of macrocycles. This review aims to give a comprehensive insight into the synthesis of varieties of macrocycles bearing indole scaffold catalyzed by various transition metals that emerged in the literature over the last two decades. We hope that this review will persuade synthetic chemists to search for novel strategies for the C–C bond macrocyclization by metal-catalyzed protocols.
Full article
(This article belongs to the Collection Advanced Research Papers in Organics)
►▼
Show Figures
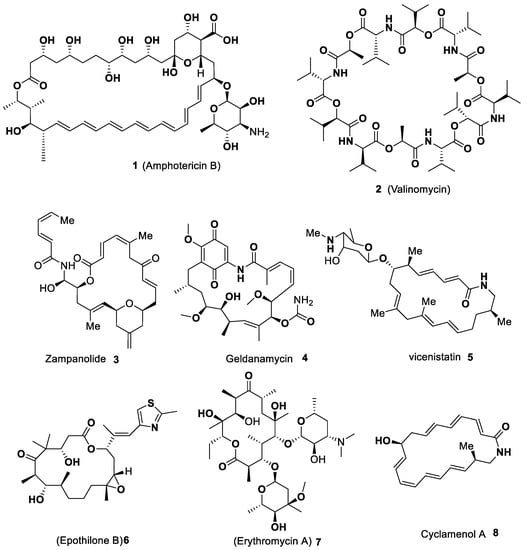
Figure 1
Open AccessArticle
Consecutive Four-Component Coupling-Addition Aza-Anellation Pictet–Spengler Synthesis of Tetrahydro-β-Carbolines: An Optimized Michael Addition and Computational Study on the Aza-Anellation Step
Organics 2023, 4(3), 313-332; https://doi.org/10.3390/org4030025 - 28 Jun 2023
Abstract
►▼
Show Figures
Starting from acid chlorides, alkynes, tryptamines, and acryloyl chloride, 21 densely substituted tetrahydro-β-carbolines were prepared in a four-component, one-pot reaction. In this study, the aza-Michael addition step to generate intermediate enaminones was optimized in the presence of ytterbium triflate. Moreover,
[...] Read more.
Starting from acid chlorides, alkynes, tryptamines, and acryloyl chloride, 21 densely substituted tetrahydro-β-carbolines were prepared in a four-component, one-pot reaction. In this study, the aza-Michael addition step to generate intermediate enaminones was optimized in the presence of ytterbium triflate. Moreover, apart from acryloyl chloride, all reactants could be deployed in almost equimolar ratios, which increases the atom economy of the sequence. For mechanistic rationalization, the concluding aza-anellation was investigated by DFT calculations on potential intermediates and corresponding activation energies, revealing that the aza-anellation proceeds via ene reaction rather than via electrocyclization.
Full article
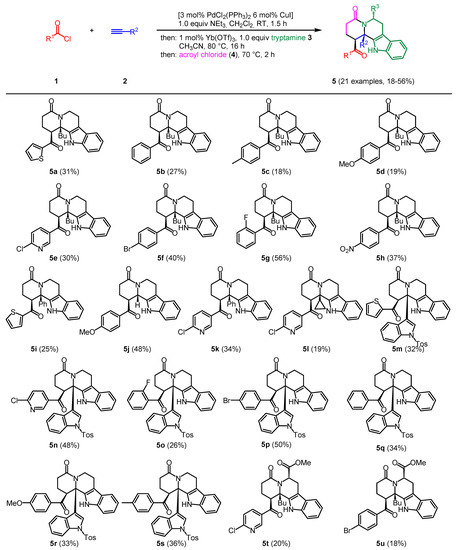
Figure 1
Open AccessArticle
Structural and Dynamic Behaviour of Heterocycles Derived from Ethylenediamines with Formaldehyde: 1,3,5-Triazinanes and Bis(imidazolidinyl)methanes
Organics 2023, 4(2), 297-312; https://doi.org/10.3390/org4020024 - 15 Jun 2023
Abstract
Formaldehyde is a simple chemical compound that is used as a building block in obtaining a wide range of products. The versatility of formaldehyde in chemical synthesis becomes evident when it is reacted with N-alkylethylenediamines. Therefore, this paper reports the structure and
[...] Read more.
Formaldehyde is a simple chemical compound that is used as a building block in obtaining a wide range of products. The versatility of formaldehyde in chemical synthesis becomes evident when it is reacted with N-alkylethylenediamines. Therefore, this paper reports the structure and reactivity of a series of compounds derived from easily accessible molecules, such as formaldehyde, sodium hydrosulphide, and N-alkylethylenediamines. The 1,3,5-triazines (1a-1d) and bis(3-alkyl-imidazolidin-1-yl)methanes (2a-2d) were obtained by simple reaction conditions. Additionally, different proportions of sodium hydrosulphide and formaldehyde were used with N-benzylamine to obtain N-benzyltriazinane (3), N-benzylthiadiazinane (4) and N-benzyldithiazinane (5). All these compounds were characterized by analytical, spectroscopic, and spectrometric techniques, such as melting point, solubility, one-dimensional and two-dimensional nuclear magnetic resonance (13C, 1H, 15N, COSY, HETCOR, NOESY, COLOC), elemental analysis, high- and low-resolution mass spectrometry, among others. The structures of compounds 4 and 5 were obtained by single-crystal X-ray diffraction. The results show that small variations in the stoichiometry and the reaction conditions significantly influence the products obtained.
Full article
(This article belongs to the Collection Advanced Research Papers in Organics)
►▼
Show Figures
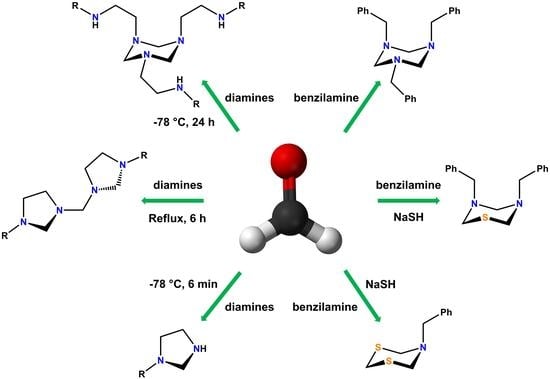
Graphical abstract
Open AccessCommunication
Palladium Catalyzed Allylic C-H Oxidation Enabled by Bicyclic Sulfoxide Ligands
Organics 2023, 4(2), 289-296; https://doi.org/10.3390/org4020023 - 13 Jun 2023
Abstract
The activation of C-H bonds is a potent tool for modifying molecular structures in chemistry. This article details the steps involved in a novel ligand bearing a bicyclic [3.3.1]-nonane framework and bissulfoxide moiety. A palladium catalyzed allylic C-H oxidation method enables a direct
[...] Read more.
The activation of C-H bonds is a potent tool for modifying molecular structures in chemistry. This article details the steps involved in a novel ligand bearing a bicyclic [3.3.1]-nonane framework and bissulfoxide moiety. A palladium catalyzed allylic C-H oxidation method enables a direct benzyl-allylic functionalization with the bissulfoxide ligand. Bissulfoixde ligand possesses a rapidly constructed bicyclic [3.3.1] framework and it proved to be effective for enabling both N- and C-alkylation. A total of 13 C-H activation productions were reported with good to excellent yields. This report validated that it is necessary to include bissulfoxide as a ligand for superior reactivities. Naftifine was produced utilizing developed C-H functionalization methodology in good overall yields.
Full article
(This article belongs to the Special Issue Design and Development of New Organic Synthetic Methods and Techniques)
►▼
Show Figures
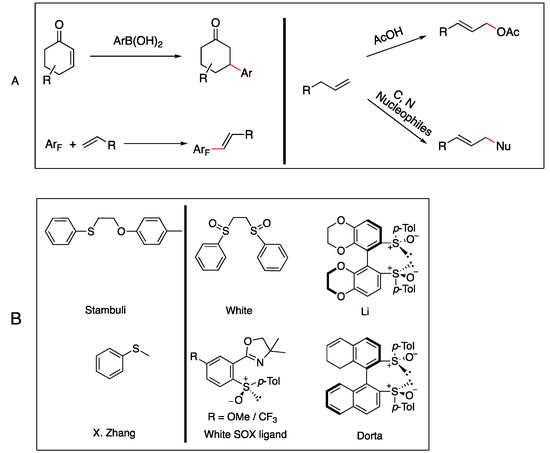
Figure 1
Open AccessReview
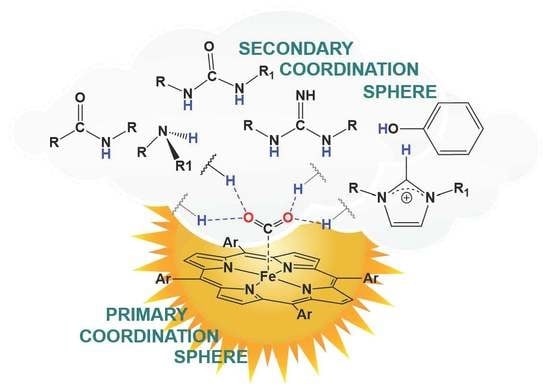
Hydrogen-Bonding Secondary Coordination Sphere Effect on CO2 Reduction
by
and
Organics 2023, 4(2), 277-288; https://doi.org/10.3390/org4020022 - 05 Jun 2023
Abstract
►▼
Show Figures
Great efforts of the scientific community are focused on the development of catalysts for the reduction of carbon dioxide (CO2) to useful molecules such as carbon monoxide, formic acid, methanol, ethanol, methane, ethylene, or acetate. Various metal porphyrin complexes were synthesized
[...] Read more.
Great efforts of the scientific community are focused on the development of catalysts for the reduction of carbon dioxide (CO2) to useful molecules such as carbon monoxide, formic acid, methanol, ethanol, methane, ethylene, or acetate. Various metal porphyrin complexes were synthesized and studied to develop highly active and selective catalysts. While the substituents on the porphyrin core (the primary coordination sphere) determine the reactivity of the metal, the introduction of the secondary coordination is important for the binding and activation of CO2. In this review, selected examples of iron porphyrin catalysts with a secondary coordination sphere capable of stabilizing intermediates of the CO2 reduction process by hydrogen bonding are presented.
Full article
Graphical abstract
Open AccessArticle
The Use of a Natural Polysaccharide as a Solidifying Agent and Color-Fixing Agent on Modern Paper and Historical Materials
Organics 2023, 4(2), 265-276; https://doi.org/10.3390/org4020021 - 02 Jun 2023
Abstract
This article presents results on the use of a new material as a solidifying agent and/or color-fixing agent. A special polysaccharide material extracted from the prickly pear cactus (Opuntia ficus indica) was tested on historical materials and modern papers. An old book from
[...] Read more.
This article presents results on the use of a new material as a solidifying agent and/or color-fixing agent. A special polysaccharide material extracted from the prickly pear cactus (Opuntia ficus indica) was tested on historical materials and modern papers. An old book from the 18th century was chosen as historical material. From the mentioned book 42 pages were taken, on which a conservation and restoration pretreatment was performed before applying the polysaccharide material: sampling, fiber analysis, dry cleaning, ink solubility, pH test, thickness measurement and wet cleaning. The paper sheets provided for the test were divided into 4 groups, 3 of which were treated with gel and one left untreated as a reference. The division into groups is not only due to the different method of application, but also due to the process of gel extraction. The effect of the treatment was analyzed using FTIR-ATR. To test the mucilage as color-fixing agent 2 samples were prepared using watercolor papers colored with 6 different watercolors applied to 2.5 cm2 samples for each color in two rows of different intensity. One of the 2 samples was treated with gel, but both were immersed for 3 times in a water-ethanol solution for approximately 20 min as is standard practice in conservation and restoration. After washing, both specimens were subjected to colorimetric analysis to assess their differences. The results provided satisfactory evidence for the protection of paints sensitive to aqueous treatments and suggest the use of mucilage as a new material in cleaning method for water-soluble media.
Full article
(This article belongs to the Collection Advanced Research Papers in Organics)
►▼
Show Figures

Figure 1
Open AccessArticle
Facile Photochemical/Thermal Assisted Hydration of Alkynes Catalysed under Aqueous Media by a Chalcogen Stabilized, Robust, Economical, and Reusable Fe3Se2(CO)9 Cluster
by
, , , and
Organics 2023, 4(2), 251-264; https://doi.org/10.3390/org4020020 - 25 May 2023
Abstract
In this report, the catalytic potential of chalcogen-stabilized iron carbonyl clusters [Fe3E2(CO)9 (E = S, Se, Te)] for the photolytic hydration of alkynes has been explored. The iron chalcogenide clusters bring excellent transformations of terminal and internal alkynes
[...] Read more.
In this report, the catalytic potential of chalcogen-stabilized iron carbonyl clusters [Fe3E2(CO)9 (E = S, Se, Te)] for the photolytic hydration of alkynes has been explored. The iron chalcogenide clusters bring excellent transformations of terminal and internal alkynes to their respective keto products in just 25 min photolysis at −5 °C in inert free and aqueous conditions. After the completion of the reaction, the product can be extracted from organic solvent, and due to the lower solubility of the catalyst in water, it can also be isolated and further reused several times prior to any activation. The catalyst was also found to be active in thermal conditions and bring about the desired transformations with average to good catalytic efficiency. Moreover, during the thermal reaction, the catalyst decomposed and formed the nanoparticles of iron selenides, which worked as a single-source precursor for FeSe nanomaterials. The presented photolysis methodology was found to be most feasible, economical, instantly produce the desired product, and work for a wide range of internal and terminal alkynes; hence, all these features made this method superior to the other reported ones. This report also serves as the first catalytic report of chalcogen-stabilized iron carbonyl clusters for alkyne hydrations.
Full article
(This article belongs to the Collection Advanced Research Papers in Organics)
►▼
Show Figures
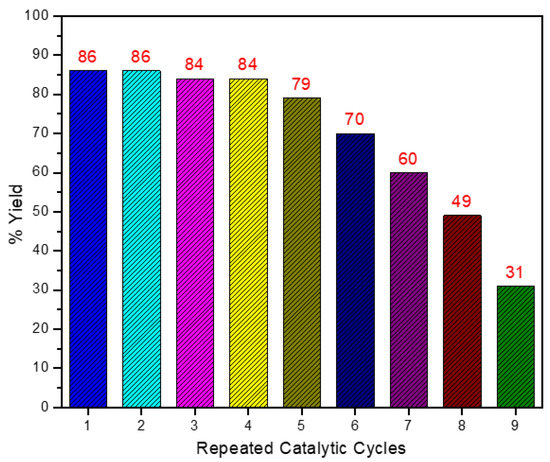
Figure 1
Open AccessReview
Synthetic Organic Molecules as Metallic Corrosion Inhibitors: General Aspects and Trends
Organics 2023, 4(2), 232-250; https://doi.org/10.3390/org4020019 - 17 May 2023
Abstract
Organic molecules are gaining special attention over the last years in the corrosion area thanks to their general low achievable cytotoxicity, structural versatility, and environmentally friendly obtainment methods. Under those approaches, synthetic organic motifs have attracted the interest of researchers due to their
[...] Read more.
Organic molecules are gaining special attention over the last years in the corrosion area thanks to their general low achievable cytotoxicity, structural versatility, and environmentally friendly obtainment methods. Under those approaches, synthetic organic motifs have attracted the interest of researchers due to their variated methods of obtention through molecular manipulation via diverse chemical reactions, allowing the production of adequately planned structures or repurposing their original application in the case of drugs. This review summarizes general aspects that are desired in organic molecules as corrosion inhibitors, presenting selected works published in the 2022–2023 period and emphasizing the importance of finding novel and different organic corrosion inhibitors. Patents were not considered in this review. Scifinder, Google Scholar, and Web of Science were employed as databases. Mathematical and analytical methods involved in the search for corrosion inhibitors are out of this review’s scope.
Full article
(This article belongs to the Collection Advanced Research Papers in Organics)
►▼
Show Figures
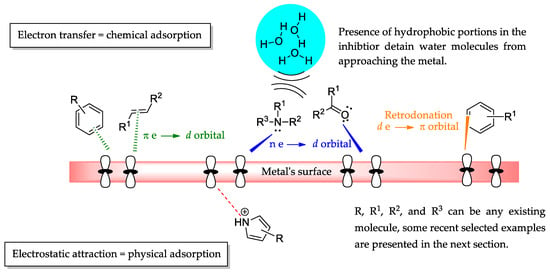
Figure 1
Open AccessArticle
A Novel Class of Functionally Tuneable Star-Shaped Molecules for Interaction with Multiple Proteins
Organics 2023, 4(2), 219-231; https://doi.org/10.3390/org4020018 - 16 May 2023
Abstract
Molecules with tuneable properties are well known for their applications in the material and bio-medical fields; nevertheless, the structural and functional tunability makes them more significant in diverse applications. Herein, we designed and synthesized a novel class of star-shaped molecules via incorporating two
[...] Read more.
Molecules with tuneable properties are well known for their applications in the material and bio-medical fields; nevertheless, the structural and functional tunability makes them more significant in diverse applications. Herein, we designed and synthesized a novel class of star-shaped molecules via incorporating two important functional groups, i.e., triazole and dithiocarbamate (DTC). The rationale behind selecting these two key functional groups is their diverse applications, e.g., DTC having applications for therapeutics, pesticides, and vulcanizing agents, and triazole having applications for anti-cancer, fungicides, anti-microbials, inhibitors, etc. The structure of the molecules was strategically designed in such a way that their overall structures are the same (central tertiary-amine and peripheral hydroxy groups), except the key functional group (DTC and triazole) in the respective molecules was different. Following synthesis and characterization, the influence of DTC and triazole groups on their bioactivity was compared via interacting with the most abundant proteins present in the blood, including serum albumin, trypsin, haemoglobin, and ribonuclease. From both the experimental and molecular docking studies, it was confirmed that the triazole molecule has a higher binding affinity towards these proteins as compared to the DTC molecule. In summary, two star-shaped DTC- and triazole-based molecules were synthesized and their bioactivity was compared via binding with blood plasma proteins.
Full article
(This article belongs to the Collection Advanced Research Papers in Organics)
►▼
Show Figures
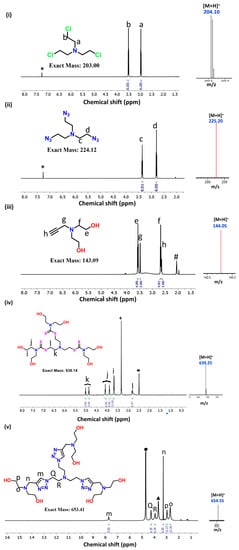
Figure 1
Open AccessArticle
The Regio- and Stereoselective Synthesis of 1,4-Diarylbut-1-en-3-ynes Having Aryl Groups at the Mutual Syn Positions
Organics 2023, 4(2), 206-218; https://doi.org/10.3390/org4020017 - 08 May 2023
Abstract
►▼
Show Figures
(E)-1-aryl-2,4-bis(trimethylsilyl)but-1-en-3-ynes readily undergo protodesilylation and subsequent aerobic, copper-free Sonogashira cross-coupling with aryl halides to form (E)-1,4-diaryl-2-(trimethylsilyl)but-1-en-3-ynes. The proposed one-pot, two-step approach allows access to the isomers containing aryl substituents in mutual syn positions. The resulting 2-silyl enynes can be further converted by proto-
[...] Read more.
(E)-1-aryl-2,4-bis(trimethylsilyl)but-1-en-3-ynes readily undergo protodesilylation and subsequent aerobic, copper-free Sonogashira cross-coupling with aryl halides to form (E)-1,4-diaryl-2-(trimethylsilyl)but-1-en-3-ynes. The proposed one-pot, two-step approach allows access to the isomers containing aryl substituents in mutual syn positions. The resulting 2-silyl enynes can be further converted by proto- or halodesilylation.
Full article
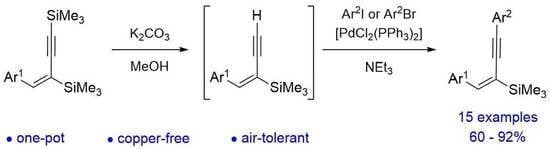
Graphical abstract
Open AccessArticle
Computational Modeling of the Feasibility of Substituted [1.1.1]Propellane Formation from Anionic Bridgehead Bromide Precursors
by
, , , , , , , , , , , , , and
Organics 2023, 4(2), 196-205; https://doi.org/10.3390/org4020016 - 04 May 2023
Cited by 1
Abstract
[1.1.1]Propellane, a compound whose structure includes two saturated carbons in which all four bonds are directed into a single hemisphere, is of theoretical interest, but has also seen recent practical applications. Mono-, di-, and trisubstituted derivatives of this propellane (by substitution of its
[...] Read more.
[1.1.1]Propellane, a compound whose structure includes two saturated carbons in which all four bonds are directed into a single hemisphere, is of theoretical interest, but has also seen recent practical applications. Mono-, di-, and trisubstituted derivatives of this propellane (by substitution of its CH2 bridges with O, S, NH, CF2, CO, SO, and SO2) remain unknown despite several computational studies that have suggested some may be stable. In this study, we show that, in several cases, substituted propellanes are spontaneously formed upon the attempted computational optimization of the geometries of anionic bridgehead bromide precursors using the ωB97X-D/aug-cc-pVDZ DFT method. Spontaneous formation suggests that these propellanes are at lower energy relative to the precursors and, therefore, are promising synthetic targets. The success or failure to spontaneously form the propellane is considered in relation to the length and strain energy of the central bridgehead-bridgehead bond, as well as the total strain energy of each propellane.
Full article
(This article belongs to the Collection Advanced Research Papers in Organics)
►▼
Show Figures
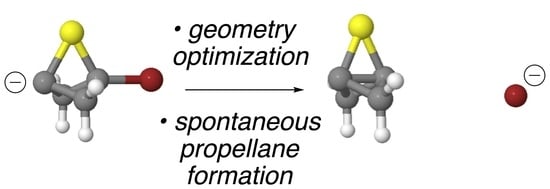
Graphical abstract
Open AccessCommunication
A Reaction of N-Substituted Succinimides with Hydroxylamine as a Novel Approach to the Synthesis of Hydroxamic Acids
Organics 2023, 4(2), 186-195; https://doi.org/10.3390/org4020015 - 27 Apr 2023
Abstract
We describe a novel two-step approach for the synthesis of compounds with a hydroxyl-amide group (hydroxamic acids), which are widely known for their biological activity (histone deacetylase inhibitors, matrix metalloproteinases inhibitors and others). The first stage is the synthesis of N-substituted succinimide
[...] Read more.
We describe a novel two-step approach for the synthesis of compounds with a hydroxyl-amide group (hydroxamic acids), which are widely known for their biological activity (histone deacetylase inhibitors, matrix metalloproteinases inhibitors and others). The first stage is the synthesis of N-substituted succinimide via the reaction of aromatic amine or carboxylic acid hydrazide with succinic anhydride. The second step involves the imide ring opening reaction by hydroxylamine. For both stages, universal synthetic methods are developed to exclude additional purification procedures for the target compounds. Sixteen hydroxamic acids are synthesized using the developed approach. Most of the compounds are obtained for the first time.
Full article
(This article belongs to the Special Issue Design and Development of New Organic Synthetic Methods and Techniques)
►▼
Show Figures
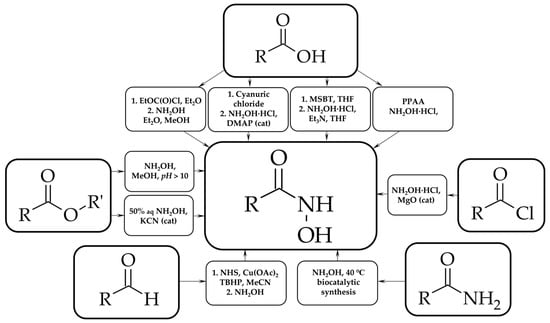
Figure 1
Open AccessReview
Advance in the Synthesis of Sulfoxides and Sulfinamides from β-Sulfinyl Esters
Organics 2023, 4(2), 173-185; https://doi.org/10.3390/org4020014 - 19 Apr 2023
Abstract
Sulfoxides and sulfinamides play important roles in the pharmaceutical industry, organic synthesis and fine chemicals. This review will demonstrate that, under catalysis by transition metals, β-sulfinyl esters, as nucleophilic reagents, react with a variety of electrophilic reagents to produce sulfoxides and sulfinamides.
[...] Read more.
Sulfoxides and sulfinamides play important roles in the pharmaceutical industry, organic synthesis and fine chemicals. This review will demonstrate that, under catalysis by transition metals, β-sulfinyl esters, as nucleophilic reagents, react with a variety of electrophilic reagents to produce sulfoxides and sulfinamides. The important prospect of the asymmetric catalytic synthesis of chiral sulfur-containing molecules in this field is described.
Full article
(This article belongs to the Collection Advanced Research Papers in Organics)
►▼
Show Figures
Figure 1
Open AccessArticle
Reinvestigation of the Room Temperature Photochemical Reaction between N-Methyl-1,2,4-triazoline-3,5-dione (MeTAD) and Benzene
Organics 2023, 4(2), 164-172; https://doi.org/10.3390/org4020013 - 17 Apr 2023
Abstract
The photochemical reaction of N-methyl-1,2,4-triazoline-3,5-dione (MeTAD) with benzene is known to lead to a Diels–Alder cycloaddition product when conducted at low temperatures (i.e., <−60 °C). This reactivity has been exploited recently for novel synthetic applications. It was previously reported that no reaction
[...] Read more.
The photochemical reaction of N-methyl-1,2,4-triazoline-3,5-dione (MeTAD) with benzene is known to lead to a Diels–Alder cycloaddition product when conducted at low temperatures (i.e., <−60 °C). This reactivity has been exploited recently for novel synthetic applications. It was previously reported that no reaction between MeTAD and benzene occurs at room temperature. However, it has now been discovered that MeTAD reacts effectively with benzene upon visible light irradiation over a several day period at room temperature. The major product is a para-substituted bisurazole adduct. Our studies indicate that the adduct is formed via sequential aromatic substitution reactions made possible by electron transfer from the aromatic ring to the highly electrophilic triplet state of photoactivated MeTAD.
Full article
(This article belongs to the Special Issue Chemistry of Heterocycles)
►▼
Show Figures
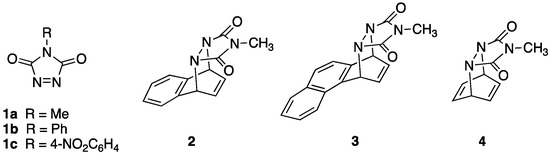
Figure 1
Open AccessArticle
Exploration of the Divergent Outcomes for the Nenitzescu Reaction of Piperazinone Enaminoesters
Organics 2023, 4(2), 146-163; https://doi.org/10.3390/org4020012 - 07 Apr 2023
Abstract
The Nenitzescu reaction is a condensation reaction between an enamine and a quinone, which can give rise to a wide variety of reaction products depending on the nature of the starting material and the reaction conditions. The most commonly observed products are 5-hydroxyindoles
[...] Read more.
The Nenitzescu reaction is a condensation reaction between an enamine and a quinone, which can give rise to a wide variety of reaction products depending on the nature of the starting material and the reaction conditions. The most commonly observed products are 5-hydroxyindoles and 5-hydroxybenzofurans. Both classes are of interest since they are known to possess a variety of promising bioactivities. Despite the high chemodivergency for this reaction, it remains an interesting synthetic strategy thanks to the mild reaction conditions, easily accessible starting materials and simple reaction procedures. For these reasons, our research group investigated the Nenitzescu reaction of piperazinone enaminoesters, resulting in the unexpected formation of rearranged 2-imidazolidinone benzofurans. In this work, we aimed to develop reaction conditions that favor the formation of 5-hydroxyindoles via an extensive, multivariate optimization study. This led to valuable insights into the parameters that influence regio- and chemoselectivity. Furthermore, two novel products were obtained, a pyrrolo[2,3-f]indole and a benzofuranone, both of which are rarely reported in the literature.
Full article
(This article belongs to the Special Issue Chemistry of Heterocycles)
►▼
Show Figures
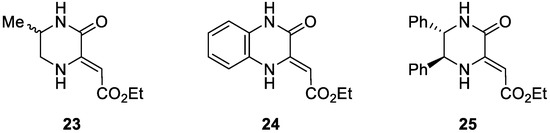
Figure 1
Open AccessCommunication
A New Rapid and Specific Iodination Reagent for Phenolic Compounds
Organics 2023, 4(2), 137-145; https://doi.org/10.3390/org4020011 - 04 Apr 2023
Abstract
►▼
Show Figures
A new rapid iodination reagent, N1,N3,N5-tris[(2,4,6-trimethylpyridine)iodo(I)]-2,4,6-triphenyl-s-triazine trihexafluorophosphate, was synthesized in a modification of the established synthesis of 2,4,6-triiodo-3,5-dimethylphenol in the presence of bis(2,4,6-trimethylpyridine)iodo(I) hexafluorophosphate and used for the precise post-modification of mono- and trisubstituted
[...] Read more.
A new rapid iodination reagent, N1,N3,N5-tris[(2,4,6-trimethylpyridine)iodo(I)]-2,4,6-triphenyl-s-triazine trihexafluorophosphate, was synthesized in a modification of the established synthesis of 2,4,6-triiodo-3,5-dimethylphenol in the presence of bis(2,4,6-trimethylpyridine)iodo(I) hexafluorophosphate and used for the precise post-modification of mono- and trisubstituted phenyl compounds. We performed triple iodinations with our new phenyl-based compounds as a proof of principle of selected types of phenols, ß-sympatholytic agents and their spin-labeled derivatives, which can be employed in electron paramagnetic resonance (EPR) spectroscopy. The new rapid iodination reagent can be employed with high reactivity and regioselectivity.
Full article
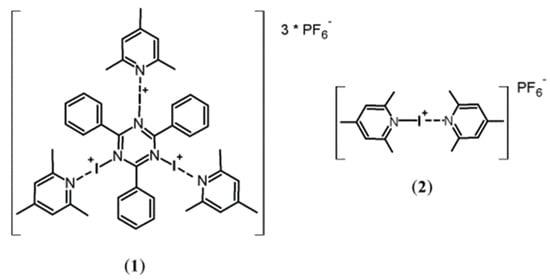
Figure 1
Highly Accessed Articles
Latest Books
E-Mail Alert
News
Topics

Conferences
Special Issues
Special Issue in
Organics
The Modern Trends in Organic Synthesis, including PASE and Green Chemistry
Guest Editors: Denis N. Bazhin, Yulia S. KudyakovaDeadline: 31 August 2023
Special Issue in
Organics
Redox Transformations in Advanced Organic Synthesis
Guest Editor: Ioannis LykakisDeadline: 30 September 2023
Special Issue in
Organics
Design and Development of New Organic Synthetic Methods and Techniques
Guest Editors: Ting Wang, Li XiaoDeadline: 31 October 2023
Special Issue in
Organics
Aromatic Heterocycles: A Wonderful Pool of Organic Materials
Guest Editor: Filip BurešDeadline: 31 December 2023
Topical Collections
Topical Collection in
Organics
Advanced Research Papers in Organics
Collection Editors: Wim Dehaen, Michal Szostak, Huaping Xu