Journal Description
Inorganics
Inorganics
is an international, scientific, peer-reviewed, open access journal of inorganic chemistry published monthly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, SCIE (Web of Science), CAPlus / SciFinder, and other databases.
- Journal Rank: JCR - Q2 (Chemistry, Inorganic & Nuclear) / CiteScore - Q2 (Inorganic Chemistry)
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 13 days after submission; acceptance to publication is undertaken in 2.9 days (median values for papers published in this journal in the first half of 2023).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
- Testimonials: See what our authors say about Inorganics.
Impact Factor:
2.9 (2022);
5-Year Impact Factor:
2.5 (2022)
Latest Articles
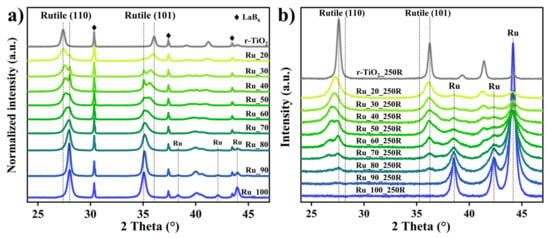
Hydrogen Incorporation in RuxTi1−xO2 Mixed Oxides Promotes Total Oxidation of Propane
Inorganics 2023, 11(8), 330; https://doi.org/10.3390/inorganics11080330 - 07 Aug 2023
Abstract
A rational synthetic approach is introduced to enable hydrogen insertion into oxides by forming a solid solution of a reducible oxide with a less reducible oxide as exemplified with RuO2 and TiO2 (Ru_x, a mixture of x% RuO2 with (100−x)%
[...] Read more.
A rational synthetic approach is introduced to enable hydrogen insertion into oxides by forming a solid solution of a reducible oxide with a less reducible oxide as exemplified with RuO2 and TiO2 (Ru_x, a mixture of x% RuO2 with (100−x)% TiO2). Hydrogen exposure at 250 °C to Ru_x (Ru_x_250R) results in substantial hydrogen incorporation accompanied by lattice strain that in turn induces pronounced activity variations. Here, we demonstrate that hydrogen incorporation in mixed oxides promotes the oxidation catalysis of propane combustion with Ru_60_250R being the catalytically most active catalyst.
Full article
(This article belongs to the Special Issue 10th Anniversary of Inorganics: Inorganic Materials)
►
Show Figures
Open AccessArticle
Influence of Ga Substitution on the Local Structure and Luminescent Properties of Eu-Doped CaYAlO4 Phosphors
Inorganics 2023, 11(8), 329; https://doi.org/10.3390/inorganics11080329 - 06 Aug 2023
Abstract
Understanding the local environment of luminescent centers in phosphors serves as a blueprint for designing the luminescent properties of phosphors. Chemical substitution is a general strategy for engineering the local structure around luminescent center ions. In this study, we systematically investigate the luminescent
[...] Read more.
Understanding the local environment of luminescent centers in phosphors serves as a blueprint for designing the luminescent properties of phosphors. Chemical substitution is a general strategy for engineering the local structure around luminescent center ions. In this study, we systematically investigate the luminescent properties of Ga-substituted Eu-doped CaYAlO4 (CYAGO:Eu) phosphors and the local structure of the Eu ions. The Ga substitution at the Al sites leads to a significant enhancement in the electric dipole transition of Eu3+ (5D0 → 7F2). The Judd–Ofelt analysis reveals that Eu3+ ions are substituted for Ca/Y, and the Ga substitution increases the asymmetricity of the local structure around the Eu ions because of the different ionic radii and electronegativities of Al and Ga. In addition, Eu2+ emission is missing regardless of the Ga substitution and post-hydrogen treatments. The present work provides deeper insight into the role of chemical substitution in oxide phosphors.
Full article
(This article belongs to the Section Inorganic Solid-State Chemistry)
►▼
Show Figures

Figure 1
Open AccessArticle
Phytochemical Substances—Mediated Synthesis of Zinc Oxide Nanoparticles (ZnO NPS)
Inorganics 2023, 11(8), 328; https://doi.org/10.3390/inorganics11080328 - 05 Aug 2023
Abstract
►▼
Show Figures
Artemisia absinthium (A. absinthium) leaf extract was successfully used to create zinc oxide nanoparticles (ZnO NPs), and their properties were investigated via several techniques, including X-ray diffraction (XRD), scanning electron microscopy (SEM), energy-dispersive X-ray (EDX), Fourier transform infrared (FTIR), and ultraviolet–visible
[...] Read more.
Artemisia absinthium (A. absinthium) leaf extract was successfully used to create zinc oxide nanoparticles (ZnO NPs), and their properties were investigated via several techniques, including X-ray diffraction (XRD), scanning electron microscopy (SEM), energy-dispersive X-ray (EDX), Fourier transform infrared (FTIR), and ultraviolet–visible spectroscopy (UV–Vis spectroscopy). SEM analysis confirmed the spherical and elliptical shapes of the particles. Three different zinc peaks were observed via EDX at the energies of 1, 8.7, and 9.8 keV, together with a single oxygen peak at 0.5 keV. The XRD analysis identified ZnO NPs as having a hexagonal wurtzite structure, with a particle size that decreased from 24.39 to 18.77 nm, and with an increasing surface area (BET) from 4.003 to 6.032 m2/g for the ZnO (without extract) and green ZnO NPs, respectively. The FTIR analysis confirmed the groups of molecules that were accountable for the stabilization and minimization of the ZnO NPs, which were apparent at 3400 cm. Using UV–Vis spectroscopy, the band-gap energies (Egs) for the green ZnO and ZnO (without extract) NPs were estimated, and the values were 2.65 and 2.79 eV, respectively.
Full article

Figure 1
Open AccessArticle
A Series of Lanthanide Complexes with Keggin-Type Monolacunary Phosphotungstate: Synthesis and Structural Characterization
Inorganics 2023, 11(8), 327; https://doi.org/10.3390/inorganics11080327 - 01 Aug 2023
Abstract
The coordination of rare-earth metal ions (Ln3+) to polyoxometalates (POM) is regarded as a way of modifying and controlling their properties, such as single-molecular magnetism or luminescent behavior. The half-sandwich complexes of Ln3+ with monolacunary Keggin POMs (Ln3+/POM
[...] Read more.
The coordination of rare-earth metal ions (Ln3+) to polyoxometalates (POM) is regarded as a way of modifying and controlling their properties, such as single-molecular magnetism or luminescent behavior. The half-sandwich complexes of Ln3+ with monolacunary Keggin POMs (Ln3+/POM = 1:1) are of particular interest, since the Ln3+ retains its ability to coordinate extra ligands. Thus, the knowledge of the exact structures of 1:1 Ln/POM complexes is important for the development of reliable synthetic protocols for hybrid complexes. In this work, we isolated three 1:1 Gd3+/POM complexes of the general formula Cat4Gd(PW11O39)·xH2O (Cat = K+ or Me4N+). Complex (Me4N)2K2[Gd(H2O)2PW11O39]·5H2O (1) is polymeric, revealing a layered structural motif via bridging Gd3+ and K+ ions. Complexes (Me4N)6K2[Gd(H2O)3PW11O39]2·20H2O (2) and (Me4N)7K[Gd(H2O)3PW11O39]2·12H2O (3) are classified as dimeric; the difference between them consists of the different crystal packing of the polyoxometalates, which is induced by a variation in the cationic composition. Isostructural complexes have also been characterized for praseodymium, europium, terbium and dysprosium. The coordination number of Ln3+ (8) persists in all the compounds, while the binding mode of the POM varies, giving rise to different architectures with two or three H2O co-ligands per Ln3+. However, whatever the particular structure and exact composition, the {Ln(PW11O39)} moieties are always involved in bonding with each other with the formation either of polymeric chains or dimeric units. In water, these aggregates can dissociate with the formation of [Ln(H2O)4PW11O39]4-. This behavior must be taken into account when choosing L for the design of hybrid {Ln(L)POM} complexes.
Full article
(This article belongs to the Special Issue Advances in Polyoxometalates for Supramolecular Architecture, Biomimetics and Bioapplications)
►▼
Show Figures

Figure 1
Open AccessArticle
PEPPSI-Type Pd(II)—NHC Complexes on the Base of p-tert-Butylthiacalix[4]arene: Synthesis and Catalytic Activities
by
, , , , , , , and
Inorganics 2023, 11(8), 326; https://doi.org/10.3390/inorganics11080326 - 01 Aug 2023
Abstract
The creation of effective catalytic systems for cross-coupling reactions, reduction, etc., capable of working in water-organic or pure aqueous media is in great demand. The article presents the synthesis of NHC-palladium complexes of the PEPPSI type based on monoimidazolium derivatives of thiacalix[4]arene. The
[...] Read more.
The creation of effective catalytic systems for cross-coupling reactions, reduction, etc., capable of working in water-organic or pure aqueous media is in great demand. The article presents the synthesis of NHC-palladium complexes of the PEPPSI type based on monoimidazolium derivatives of thiacalix[4]arene. The structure of the imidazolium precursors, obtained in 81–88% yields and the complexes themselves, obtained in 40–50% yields, is established using modern methods, including X-ray structural analysis and high-resolution mass spectrometry. It is shown that the obtained complex with bulk substituents near the palladium atom is not inferior to the well-known PEPPSI-type Organ’s catalyst in the catalysis of Suzuki-Miyaura coupling and is four times superior to the latter in the p-nitrophenol reduction reaction. Given the presence of free phenolic hydroxyl groups in the macrocycle, the obtained complexes are of interest for further post-modification or for immobilization on a carrier.
Full article
(This article belongs to the Special Issue N-Heterocyclic Carbene Metal Complexes: Synthesis, Properties and Applications)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Study of the Cathode Pt-Electrocatalysts Based on Reduced Graphene Oxide with Pt-SnO2 Hetero-Clusters
by
, , , , , , , and
Inorganics 2023, 11(8), 325; https://doi.org/10.3390/inorganics11080325 - 31 Jul 2023
Abstract
A complex study of the structure, morphology, and electrochemical properties of the Pt20/SnO210/RGO electrocatalyst is presented. The advantage of the chemical synthesis of reduced graphene oxide (c-RGO) compared to thermal methods (t-RGO) is due to the formation of
[...] Read more.
A complex study of the structure, morphology, and electrochemical properties of the Pt20/SnO210/RGO electrocatalyst is presented. The advantage of the chemical synthesis of reduced graphene oxide (c-RGO) compared to thermal methods (t-RGO) is due to the formation of graphene plates with amorphous carbon black agglomerates and the chemical composition of the surface. The nature of the interaction between platinum and tin dioxide particles and a conclusion about the formation of heterostructures Pt-SnO2 with the surface interaction of lattices excluding the formation of hetero phases has been established. This achieves high dispersity during the formation of platinum particles without significant agglomeration and increases the electrochemical surface area (ESA) of platinum to 85 m2 g−1 vs. carbon black. In addition, the surface interaction of particles and the formation of hetero-clusters Pt-SnO2 can cause the improved activity and stability of the Pt20/SnO210/c-RGO electrocatalyst.
Full article
(This article belongs to the Special Issue 10th Anniversary of Inorganics: Inorganic Solid State Chemistry)
►▼
Show Figures

Figure 1
Open AccessArticle
Oxidative, Genotoxic and Cytotoxic Damage Potential of Novel Borenium and Borinium Compounds
Inorganics 2023, 11(8), 324; https://doi.org/10.3390/inorganics11080324 - 31 Jul 2023
Abstract
In this study, the biological properties of novel borenium and borinium compounds in terms of their oxidative, genotoxic, and cytotoxic effects were assessed on cultured human peripheral blood cells, as well as several types of cancer cells. Our results revealed that the borinium
[...] Read more.
In this study, the biological properties of novel borenium and borinium compounds in terms of their oxidative, genotoxic, and cytotoxic effects were assessed on cultured human peripheral blood cells, as well as several types of cancer cells. Our results revealed that the borinium compounds yielded the best results in terms of supporting total antioxidant capacity (TAC). In fact, borenium 1, borenium 2, borenium 3, borinium 4, and borinium 5 compounds elevated TAC levels of cultured human blood cells at rates of 42.8%, 101.5%, 69.8%, 33.3%, and 49.2%, respectively. There were no statistically significant differences (p > 0.05) between the negative control and the groups treated with all borinium and borenium concentrations from the micronucleus (MN) and chromosome aberration (CA) assays, demonstrating the non-genotoxic effects. Moreover, borenium 1 (60.7% and 50.7%), borenium 2 (70.4% and 57.2%), borenium 3 (53.1% and 45.2%), borinium 4 (55.1% and 48.1%), and borinium 5 (51.0% and 36.1%) minimized the mitomycin C(MMC)-induced genotoxic damages at different rates as determined using CA and MN assays, respectively. Again, it was found that the borinium compounds exhibited higher cytotoxic activity on cancer cells when compared to borenium compounds. Consequently, in light of our in vitro findings, it was suggested that the novel borinium and borenium compounds could be used safely in pharmacology, cosmetics, and various medical fields due to their antioxidant and non-genotoxic features, as well as their cytotoxicity potential on cancer cells.
Full article
(This article belongs to the Special Issue Synthesis and Innovative Biological Activity of Boron-Containing Compounds)
►▼
Show Figures
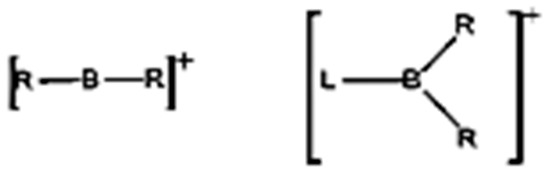
Figure 1
Open AccessArticle
[email protected]82: Monometallic Clusterfullerene Encapsulating a Tm3+ Ion
by
, , , , , and
Inorganics 2023, 11(8), 323; https://doi.org/10.3390/inorganics11080323 - 31 Jul 2023
Abstract
Metal cyanide clusterfullerenes (CYCFs) are formed via the encapsulation of a single metal atom and a cyanide unit inside fullerene cages, endowing them with excellent properties in various applications. In this work, we report the synthesis, isolation, and characterizations of the first cases
[...] Read more.
Metal cyanide clusterfullerenes (CYCFs) are formed via the encapsulation of a single metal atom and a cyanide unit inside fullerene cages, endowing them with excellent properties in various applications. In this work, we report the synthesis, isolation, and characterizations of the first cases of thulium (Tm)-based CYCFs with the popular C82 carbon cages. The structural elucidation of the two [email protected]82 isomers was achieved via diverse analytical techniques, including mass spectrometry, Vis-NIR spectroscopy, single-crystal X-ray crystallography, and cyclic voltammetry. The crystallographic analyses unambiguously confirmed the molecular structures of the two [email protected]82 isomers as [email protected]s(6)-C82 and [email protected]2v(9)-C82. Both TmCN clusters adopt a well-established triangular configuration, with the Tm ion located on the symmetrical plane of the carbon cages. The electronic structures of both [email protected]82 isomers adopt a Tm3+(CN)−@(C82)2− configuration, exhibiting characteristic spectral and electrochemical properties reminiscent of divalent endohedral metallofullerenes (EMFs). Intriguingly, unlike the divalent Tm2+ ion observed in the mono-metallofullerenes [email protected]2n, a higher oxidation state of Tm3+ is identified in the monometallic TmCN cluster due to bonding with the cyanide anion. This result provides valuable insight into the essential role of the non-metallic endo-units in governing the oxidation state of the metal ion and the electronic behaviors of EMFs.
Full article
(This article belongs to the Special Issue Research on Metallofullerenes)
►▼
Show Figures
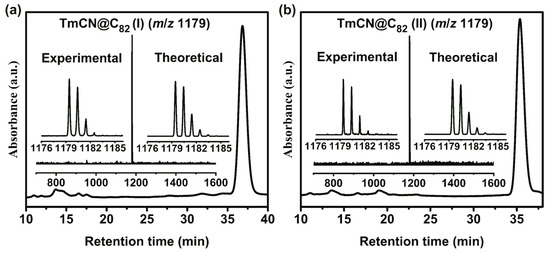
Figure 1
Open AccessArticle
Investigation of In Vitro Anticancer and Apoptotic Potential of Biofabricated Silver Nanoparticles from Cardamine hirsuta (L.) Leaf Extract against Caco-2 Cell Line
by
, , , , , , , and
Inorganics 2023, 11(8), 322; https://doi.org/10.3390/inorganics11080322 - 31 Jul 2023
Abstract
Green nanoparticle (NPs) synthesis is eco-friendly, non-toxic, and the NPs have demonstrated improved biocompatibility for use in healthcare. This study evaluated the biogenic synthesis of AgNPs from the leaves of Cardamine hirsuta L. and their biological properties. The UV-Vis. spectra at 411 nm
[...] Read more.
Green nanoparticle (NPs) synthesis is eco-friendly, non-toxic, and the NPs have demonstrated improved biocompatibility for use in healthcare. This study evaluated the biogenic synthesis of AgNPs from the leaves of Cardamine hirsuta L. and their biological properties. The UV-Vis. spectra at 411 nm exhibited a distinct resonance spectrum for C-AgNPs produced from C. hirsuta L. FT-IR analysis exhibited the presence of functional groups of phyto-compounds of C. hirsuta responsible of silver salt reduction and capping agents of C-AgNPs. The microscopic-based study, such as HR-TEM analysis, showed that the particles were uniformly distributed, spherical, and ranged in size from 5.36 to 87.65 nm. EDX analysis confirmed a silver (Ag) content of 36.3% by weight, and XRD analysis exhibited the face-centred cubic (FCC) crystalline nature of C-AgNPs. DLS measured the mean particle size of 76.5 nm. The zeta potential was significant at −27.9 mV, and TGA analysis revealed that C-AgNPs had higher thermal stability. C-AgNPs demonstrated moderate antimicrobial activity against the tested pathogens. In addition, the anti-proliferative activity measured by the MTT assay on the Caco-2 cell line demonstrated decreased cell viability with increasing C-AgNPs dosage, with an IC50 concentration of 49.14 µg/mL. In addition, an Annexin-V/Propidium iodide flow cytometric study was utilized to evaluate the induction of apoptosis in cancer cells. Early and late apoptosis cell populations increased significantly compared to the untreated control. Therefore, green-synthesized C-AgNPs have significant antimicrobial and anti-proliferative abilities, making them intriguing options for future biomedical applications.
Full article
(This article belongs to the Special Issue Functional Inorganic Materials for Biomedical Application)
►▼
Show Figures

Figure 1
Open AccessArticle
Synthesis of New Organoselenium-Based Succinanilic and Maleanilic Derivatives and In Silico Studies as Possible SARS-CoV-2 Main Protease Inhibitors
by
, , , , , , and
Inorganics 2023, 11(8), 321; https://doi.org/10.3390/inorganics11080321 - 29 Jul 2023
Abstract
Herein we report the synthesis of organic selenide-based maleanilic and succinanilic acids in good yields (up to 95%). Their structural identities were elucidated by spectroscopic techniques (e.g., IR, 1H- & 13C-NMR, and MS). The ADMET analysis, molecule electrostatic potential map, DFT,
[...] Read more.
Herein we report the synthesis of organic selenide-based maleanilic and succinanilic acids in good yields (up to 95%). Their structural identities were elucidated by spectroscopic techniques (e.g., IR, 1H- & 13C-NMR, and MS). The ADMET analysis, molecule electrostatic potential map, DFT, and frontier molecular orbital were used to study the organoselenium compounds’ pharmacokinetics, drug-likeness characteristics, geometries, and chemical and electronic properties. Moreover, a molecular docking tool was employed to investigate the organic selenides’ ability to inhibit the SARS-CoV-2 Mpro target (PDB: 7BFB). Within this context, organic selenides exhibited promising binding affinities to the SARS-CoV-2 Mpro receptor in the following order (12 > 11 > 10 > 9 > 7 > 8). Furthermore, molecular dynamics simulations were also carried out for 200 ns to evaluate the exact behavior of the most active compound (12) within the Mpro binding pocket of SARS-CoV-2 compared with its co-crystallized inhibitor (Co).
Full article
(This article belongs to the Special Issue Advances in the Domain of Organometallic Antioxidant and Anticancer Agents)
►▼
Show Figures
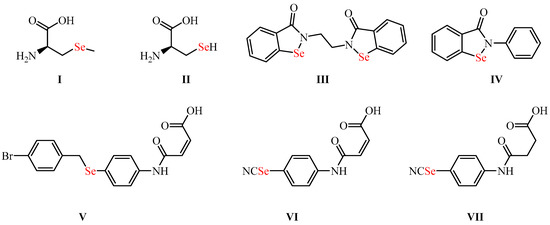
Figure 1
Open AccessReview
Metal Complexes with Schiff Bases as Antimicrobials and Catalysts
by
, , , , , , and
Inorganics 2023, 11(8), 320; https://doi.org/10.3390/inorganics11080320 - 28 Jul 2023
Abstract
Complexes of Schiff bases (SBs) with metals are promising compounds exhibiting a broad range of applications, such as catalysts, polymers, dyes, and several biological activities, including antimicrobial, anticancer, antioxidant, antimalarial, analgesic, antiviral, antipyretic, and antidiabetic actions. Considering the crisis that the whole world
[...] Read more.
Complexes of Schiff bases (SBs) with metals are promising compounds exhibiting a broad range of applications, such as catalysts, polymers, dyes, and several biological activities, including antimicrobial, anticancer, antioxidant, antimalarial, analgesic, antiviral, antipyretic, and antidiabetic actions. Considering the crisis that the whole world is now facing against antimicrobial-resistant bacteria, in the present review, we chose to focus on the activity of SBs as antimicrobials, particularly underlying the most recent studies in this field. Finally, some interesting catalytic applications recently described for metal complexes with SBs have also been discussed.
Full article
(This article belongs to the Special Issue Recent Advances in Biological and Catalytic Applications of Metal Complexes)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Tailoring of Hydrogen Generation by Hydrolysis of Magnesium Hydride in Organic Acids Solutions and Development of Generator of the Pressurised H2 Based on this Process
by
, , , , , , and
Inorganics 2023, 11(8), 319; https://doi.org/10.3390/inorganics11080319 - 27 Jul 2023
Abstract
Hydrolysis of light metals and hydrides can potentially be used for the generation of hydrogen on-board fuel cell vehicles, or, alternatively, for refilling their fuel tanks with H2 generated and pressurised without compressor on site, at near-ambient conditions. Implementation of this approach
[...] Read more.
Hydrolysis of light metals and hydrides can potentially be used for the generation of hydrogen on-board fuel cell vehicles, or, alternatively, for refilling their fuel tanks with H2 generated and pressurised without compressor on site, at near-ambient conditions. Implementation of this approach requires solution of several problems, including the possibility of controlling H2 release and avoiding thermal runaway. We have solved this problem by developing the apparatus for the controlled generation of pressurised H2 using hydrolysis of Mg or MgH2 in organic acid solutions. The development is based on the results of experimental studies of MgH2 hydrolysis in dilute aqueous solutions of acetic, citric, and oxalic acids. It was shown that the hydrogen yield approaches 100% with a fast hydrolysis rate when the molar ratio acid/MgH2 exceeds 0.9, 2.0, and 2.7 for the citric, oxalic, and acetic acids, respectively. In doing so, the pH of the reaction solutions after hydrolysis corresponds to 4.53, 2.11, and 4.28, accordingly, testifying to the buffer nature of the solutions “citric acid/magnesium citrate” and “acetic acid/magnesium acetate”. We also overview testing results of the developed apparatus where the process rate is effectively controlled by the control of the acid concentration in the hydrolysis reactor.
Full article
(This article belongs to the Special Issue Metal Hydrogen Systems: Fundamental Properties and Current Applications for Energy Storage and Conversion)
►▼
Show Figures

Figure 1
Open AccessFeature PaperArticle
Reactivity of N-Heterocyclic Stannylenes: Oxidative Addition of Chalcogen Elements to a Chiral NH-Sn System
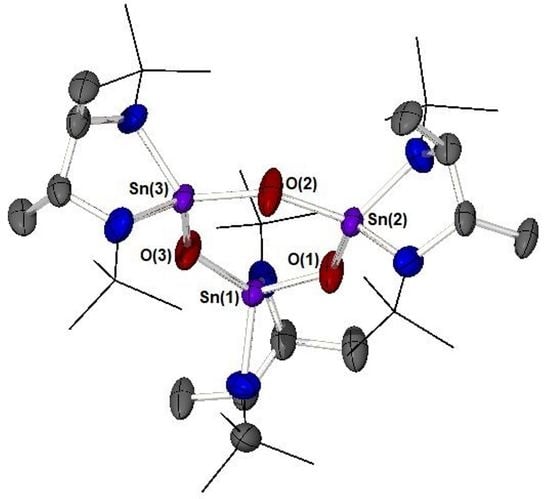
Inorganics 2023, 11(8), 318; https://doi.org/10.3390/inorganics11080318 - 27 Jul 2023
Abstract
The reactivity of the racemic N-heterocyclic stannylene [{MeHCN(tBu)}Sn] (1) with the chalcogenide elements O2, S, Se, and Te has been investigated. In the case of the reaction of 1 with molecular oxygen, the cyclic tristannoxane complex [{MeHCN(
[...] Read more.
The reactivity of the racemic N-heterocyclic stannylene [{MeHCN(tBu)}Sn] (1) with the chalcogenide elements O2, S, Se, and Te has been investigated. In the case of the reaction of 1 with molecular oxygen, the cyclic tristannoxane complex [{MeHCN(tBu)}2Sn(μ-O)]3 (3) was isolated and characterised. NMR studies (1H, 13C, and 119Sn) show the formation of D3- and C2- symmetric assemblies. The reaction of 1 with S, Se, and Te, respectively, yielded the cyclo-distannachalcogenide complexes, [{MeHCN(tBu)}2Sn(μ-E)]3 (4: E = S, 5: E = Se, 6: E = Te), again with multinuclear NMR studies proving the formation of C2- and Cs-symmetric assemblies. Single crystal X-ray diffraction studies have been used to elucidate the molecular structures of the products of oxidative addition, 3, 4, 5, and 6.
Full article
(This article belongs to the Special Issue Metal Complexes with N-donor Ligands)
►▼
Show Figures
Graphical abstract
Open AccessArticle
Photoluminescent Lanthanide(III) Coordination Polymers with Bis(1,2,4-Triazol-1-yl)Methane Linker
by
, , , and
Inorganics 2023, 11(8), 317; https://doi.org/10.3390/inorganics11080317 - 27 Jul 2023
Abstract
A series of new lanthanide(III) coordination polymers with the general formula [Ln(btrm)2(NO3)3]n, where btrm = bis(1,2,4-triazol-1-yl)methane and Ln = Eu3+, Tb3+, Sm3+, Dy3+, Gd3+ were synthesized
[...] Read more.
A series of new lanthanide(III) coordination polymers with the general formula [Ln(btrm)2(NO3)3]n, where btrm = bis(1,2,4-triazol-1-yl)methane and Ln = Eu3+, Tb3+, Sm3+, Dy3+, Gd3+ were synthesized and characterized by IR-spectroscopy, elemental, thermogravimetric, single-crystal, and powder X-ray diffraction analyses. Europium(III), samarium(III), terbium(III), and gadolinium(III) coordination polymers demonstrate thermal stability up to 250 °C, while dysprosium(III) is stable up to 275 °C. According to single-crystal X-ray diffraction analysis, the ligand exhibits a bidentate-bridging coordination mode, forming a polymeric chain of octagonal metallocycles. The photoluminescence of the free ligand in the polycrystalline state is observed in the ultraviolet range with a quantum yield of 13%. The energy transfer from the ligand to the lanthanide ions was not observed for all obtained coordination polymers. However, there are sharp bands of lanthanide(III) ions in the diffuse reflectance and excitation spectra of the obtained compounds. Therefore, Ln(III) luminescence arises, most probably, from the enhancement of f-f transition intensity under the influence of the ligand field and non-centrosymmetric interactions.
Full article
(This article belongs to the Special Issue Synthesis, Properties and Applications of Lanthanide and Actinide Molecular Compounds)
►▼
Show Figures

Figure 1
Open AccessArticle
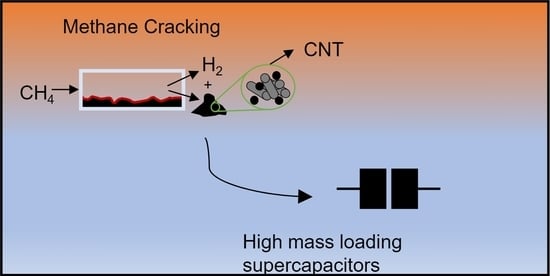
Superior Rate Capability of High Mass Loading Supercapacitors Fabricated with Carbon Recovered from Methane Cracking
by
, , , , and
Inorganics 2023, 11(8), 316; https://doi.org/10.3390/inorganics11080316 - 27 Jul 2023
Abstract
High mass loading (ca. 30 mg/cm2) electrodes were prepared with carbon recovered from catalytic methane cracking (MC). As-fabricated supercapacitors displayed 74% of capacitance retention from 6 mA/cm2 to 60 mA/cm2 and a Ragone plot’s slope of −7 Wh/kW (compared
[...] Read more.
High mass loading (ca. 30 mg/cm2) electrodes were prepared with carbon recovered from catalytic methane cracking (MC). As-fabricated supercapacitors displayed 74% of capacitance retention from 6 mA/cm2 to 60 mA/cm2 and a Ragone plot’s slope of −7 Wh/kW (compared to 42% and −31 Wh/kW, respectively, for high mass loading devices fabricated with commercial carbon). The high-rate capability of the MC-recovered carbon is attributed to the presence of carbon black and carbon nanotubes produced during the reaction, which likely increased the electronic and ionic conductivity within the electrode. These results suggest that the by-product of this hydrogen generation route might be a suitable active material for supercapacitors.
Full article
(This article belongs to the Special Issue Electrochemical Study of Nanocarbon Based Materials)
►▼
Show Figures
Graphical abstract
Open AccessArticle
Light Inorganic Scintillation Materials for Neutron and Charge Particle Detection
by
, , , , , and
Inorganics 2023, 11(8), 315; https://doi.org/10.3390/inorganics11080315 - 25 Jul 2023
Abstract
►▼
Show Figures
The technological aspects of the light inorganic crystalline compounds suitable to create scintillation materials to detect charged particles and neutrons in a wide energy range have been examined. Among them, Li2CaSiO4:Eu was found to be a prospective candidate to
[...] Read more.
The technological aspects of the light inorganic crystalline compounds suitable to create scintillation materials to detect charged particles and neutrons in a wide energy range have been examined. Among them, Li2CaSiO4:Eu was found to be a prospective candidate to control the valent state of the Rare Earth (RE) and to provide a high intensity of luminescence. It was demonstrated that the material has room for future improvement; however, this requires precise engineering of its composition—an experimental search of compositions or additives that will provide the maximum Eu2+/Eu3+ ratio to achieve a high scintillation light yield. The benefits of light inorganic materials are disclosed through the modeling of the linear density of nonequilibrium carriers along secondary particle tracks created in scintillators utilized for neutron detection. It is shown that oxide matrices have a larger linear density in comparison with halide crystalline compounds under alpha-particles and tritons, whereas light oxides can provide smaller numbers under protons.
Full article

Figure 1
Open AccessCommunication
Binder-Free CoMn2O4 Nanoflower Particles/Graphene/Carbon Nanotube Composite Film for a High-Performance Lithium-Ion Battery
Inorganics 2023, 11(8), 314; https://doi.org/10.3390/inorganics11080314 - 25 Jul 2023
Abstract
Manganese-based bimetallic oxides show a high theoretical specific capacity, making them a potential next-generation lithium-ion battery anode material. However, as with metal oxide anode materials, aggregation, volume expansion, and poor conductivity are the main obstacles. In this manuscript, flexible CoMn2O4
[...] Read more.
Manganese-based bimetallic oxides show a high theoretical specific capacity, making them a potential next-generation lithium-ion battery anode material. However, as with metal oxide anode materials, aggregation, volume expansion, and poor conductivity are the main obstacles. In this manuscript, flexible CoMn2O4/graphene/carbon nanotube films were successfully prepared through a facile filtration strategy and a subsequent thermal treatment process. When used as anodes for lithium batteries, these films can be pressed onto nickel foam without other conductive additives and binders, which simplifies the manufacturing process. When used as an anode in the lithium-ion battery, CoMn2O4/GR/CNT film exhibits a high discharge capacity of 881 mAh g−1 after 55 cycles. This value is ~2 times higher than the discharge capacity of CoMn2O4. The three-dimensional GR/CNT carrier effectively dispersed CoMn2O4, preventing its aggregation and alleviating the problem of volume expansion.
Full article
(This article belongs to the Special Issue 10th Anniversary of Inorganics: Inorganic Materials)
►▼
Show Figures
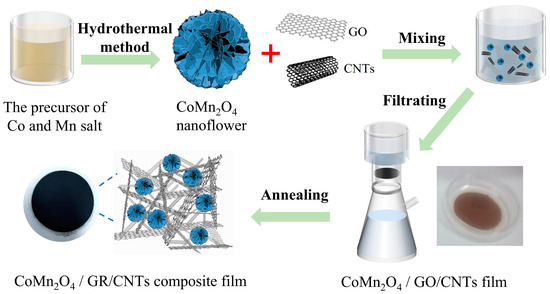
Figure 1
Open AccessArticle
[email protected]82 Metallofullerene: Calculated Isomeric Populations
Inorganics 2023, 11(7), 313; https://doi.org/10.3390/inorganics11070313 - 24 Jul 2023
Abstract
Relative equilibrium populations of the five lowest-energy isolated-pentagon-rule (IPR) isomeric structures of [email protected]
Relative equilibrium populations of the five lowest-energy isolated-pentagon-rule (IPR) isomeric structures of [email protected]
(This article belongs to the Special Issue Research on Metallofullerenes)
►▼
Show Figures
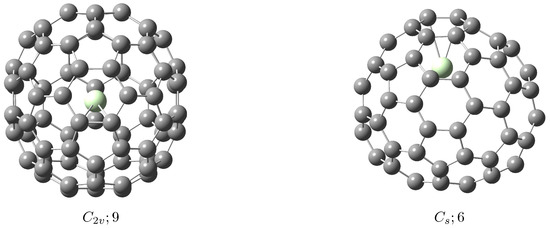
Figure 1
Open AccessArticle
Structure–Function Relationship within Cu-Peptoid Electrocatalysts for Water Oxidation
Inorganics 2023, 11(7), 312; https://doi.org/10.3390/inorganics11070312 - 24 Jul 2023
Abstract
Water oxidation (WO) is the first step in the water-splitting process aiming at the production of hydrogen as a green renewable fuel. To successfully perform WO, potent strategies for overcoming the high energetic barrier and slow kinetics of this reaction are urgently required.
[...] Read more.
Water oxidation (WO) is the first step in the water-splitting process aiming at the production of hydrogen as a green renewable fuel. To successfully perform WO, potent strategies for overcoming the high energetic barrier and slow kinetics of this reaction are urgently required. One such strategy is the use of molecular catalysis. Specifically, Cu-based catalysts have been highlighted over the last decade due to their stability and fast kinetics. Among them, Cu-peptoids, where peptoids are peptidomimetics akin to peptides and are N-substituted glycine oligomers, can act as stable and active catalysts for oxidation transformations including electrocatalytic WO. Previously, we suggested that a benzyl group incorporated as a side chain near the catalytic site within a Cu-peptoid electrocatalyst for WO has a structural role in the activity of the electrocatalyst in phosphate buffer (PBS). Herein, we aimed to test this hypothesis and understand how an incorporated structural element side chain affects WO. To this aim, we prepared a set of peptoid trimers each with a different structural element replacing the benzyl group by either naphthyl, cyclohexyl, benzyl, propyl chloride, or propyl side chains as well as a peptoid lacking a structural element. We studied the structure of their Cu complexes and tested these complexes as electrocatalysts for WO. We discovered that while all the peptoids self-assemble to form dinuclear Cu-peptoid complexes, the duplex that has no structural side chain, Cu2(BE)2, is structurally different from the others in the solid state. Moreover, Cu2(BE)2 remains dinuclear in a PBS at pH 11, while all the other duplexes are mononuclear in the PBS. Finally, though most of the complexes showed low electrocatalytic activity for WO, the dinuclear complex Cu2(BE)2 performed with the highest turnover frequency of 484 s−1. Nevertheless, this dinuclear complex slowly decomposes to the corresponding mononuclear complex as a more stable species during WO, while the other mononuclear complexes retain their structure in solution but display much slower kinetics (ca. 5 to 8 s−1) under the same conditions. Overall, our results demonstrate that bulkier side chains hamper the stability of dinuclear Cu-peptoids in a PBS, and hence, their efficiency as WO electrocatalysts is also hampered.
Full article
(This article belongs to the Special Issue Editorial Board Members’ Collection Series in “Water Oxidation Catalysts”)
►▼
Show Figures

Graphical abstract
Open AccessArticle
MoO3 Solubility and Chemical Durability of V2O5-Bearing Borosilicate Glass
by
and
Inorganics 2023, 11(7), 311; https://doi.org/10.3390/inorganics11070311 - 24 Jul 2023
Abstract
In the vitrification of high-level radioactive liquid waste (HLW), the separation of sodium-molybdate melts is a problem because it reduces the chemical durability of the vitrified waste. A glass with both high MoO3 solubility and chemical durability is required for the safe
[...] Read more.
In the vitrification of high-level radioactive liquid waste (HLW), the separation of sodium-molybdate melts is a problem because it reduces the chemical durability of the vitrified waste. A glass with both high MoO3 solubility and chemical durability is required for the safe disposal of radioactive waste. In this study, we investigate the effects of vanadium oxide on the phase separation of the molybdenum-rich phase and the water resistance of the resulting glass by phase equilibrium experiments and chemical durability test. Phase equilibrium experiments were performed on SiO2-B2O3-Al2O3-ZnO-CaO-Na2O-LiO2-MoO3 system glasses and on glasses with V2O5 added. The results showed that MoO3 solubility increased when V2O5 was added. The increase in MoO3 solubility in borosilicate melts may be associated with the viscosity-lowering effect of V2O5. Chemical durability tests were performed on borosilicate glass compositions obtained from phase equilibrium experiments. The normalized leaching rates of V2O5-bearing glasses were higher than those of other glasses. This is due to the higher network modifier/network former ratio of the glass tested. The normalized elemental mass loss of glass containing waste components increases with increasing leaching duration. This suggests that the waste component prevents the formation of a gel layer at the reaction front.
Full article
(This article belongs to the Special Issue Vanadium in the Center: Current Chemistry and Utilization of the Versatile Metal)
►▼
Show Figures

Figure 1

Journal Menu
► ▼ Journal Menu-
- Inorganics Home
- Aims & Scope
- Editorial Board
- Reviewer Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Conferences
- Editorial Office
- 10th Anniversary of Inorganics
Journal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Coatings, Inorganics, Materials, Membranes, Nanomaterials
Inorganic Thin Films and Membrane Materials
Topic Editors: Silvia Maria Deambrosis, Laura Almar, Cecilia Mortalò, Sonia EscolasticoDeadline: 30 November 2023
Topic in
Catalysts, Inorganics, IJMS, Metals, Molecules
Recent Advances in the Coordination Chemistry of Coinage Metals: From Synthesis to Optical and Biomedical Applications
Topic Editors: Artem Gushchin, Alexander Artem’evDeadline: 31 December 2023
Topic in
Catalysts, Crystals, Inorganics, Materials, Molecules
Advances in Inorganic Synthesis
Topic Editors: Andrei V. Shevelkov, Maxim N. SokolovDeadline: 31 December 2024

Conferences
Special Issues
Special Issue in
Inorganics
Modern Methods, Modern Users, and Modern Materials: Solid-State Materials Synthesis and Crystal Growth Methods
Guest Editors: W. Adam Phelan, Mariya Zhuravleva, Satya KushwahaDeadline: 25 August 2023
Special Issue in
Inorganics
Nanocomposites for Photocatalysis
Guest Editors: Alejandro Pérez-Larios, Oomman K. VargheseDeadline: 31 August 2023
Special Issue in
Inorganics
Recent Advances in Electrode Materials for Rechargeable Metal Batteries
Guest Editors: Tao Li, Long Zhang, Xue BaiDeadline: 20 September 2023
Special Issue in
Inorganics
Boron Chemistry: Fundamentals and Applications
Guest Editors: Jean-François Halet, Gilles AlcarazDeadline: 30 September 2023
Topical Collections
Topical Collection in
Inorganics
Coordination Complexes for Dye-Sensitized Solar Cells (DSCs)
Collection Editor: Catherine Housecroft